102741
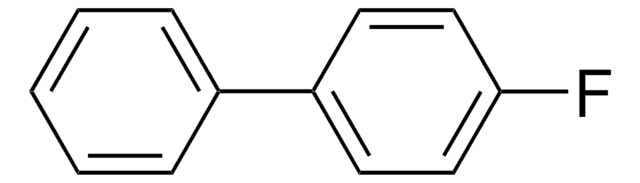
2-Fluorobiphenyl
96%
Synonym(s):
1-Fluoro-2-phenylbenzene, o-Fluorodiphenyl
About This Item
Recommended Products
Assay
96%
form
solid
bp
248 °C (lit.)
mp
71-74 °C (lit.)
functional group
fluoro
phenyl
SMILES string
Fc1ccccc1-c2ccccc2
InChI
1S/C12H9F/c13-12-9-5-4-8-11(12)10-6-2-1-3-7-10/h1-9H
InChI key
KLECYOQFQXJYBC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
US EPA Method 610 describes the analysis of polynuclear aromatic hydrocarbons (commonly referred to as PAHs or PNAs) by both HPLC and GC.
US EPA Method 8270 (Appendix IX): GC Analysis of Semivolatiles on Equity®-5 (30 m x 0.25 mm I.D., 0.50 μm)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service