86320
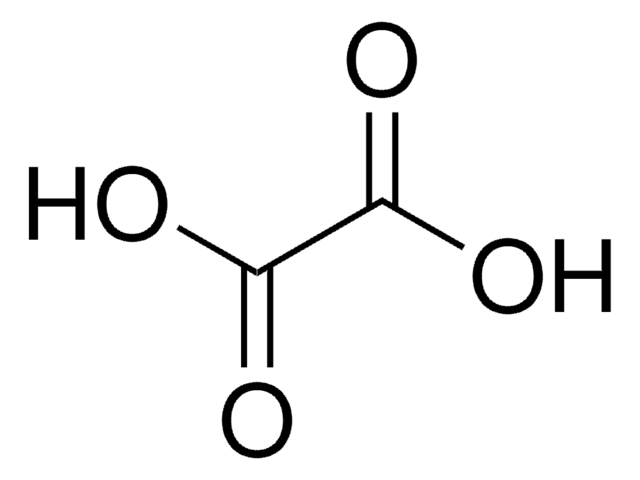
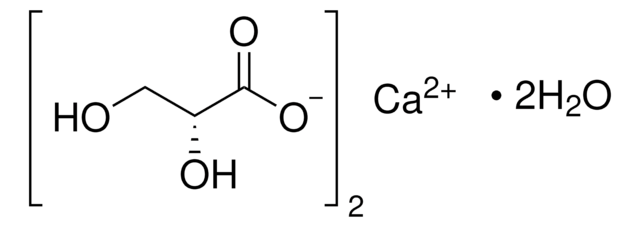
Tartronic acid
≥97.0%
Synonym(s):
Hydroxymalonic acid, Hydroxypropanedioic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
HOCH(COOH)2
CAS Number:
Molecular Weight:
120.06
Beilstein:
1209791
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥97.0%
form
powder
mp
158-160 °C (dec.) (lit.)
storage temp.
2-8°C
SMILES string
OC(C(O)=O)C(O)=O
InChI
1S/C3H4O5/c4-1(2(5)6)3(7)8/h1,4H,(H,5,6)(H,7,8)
InChI key
ROBFUDYVXSDBQM-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
- Polymer synthesis for enhanced thermal conductivity: Tartronic acid is used to exploit enzyme reactions in polymer synthesis, significantly increasing the thermal conductivity of materials, which is pivotal in manufacturing and material science applications (Nan et al., 2023).
- Advances in green chemical treatments: This acid plays a role in the electro-oxidation pathways for treating glycerol waste, contributing to sustainable chemical processes and green chemistry applications, which are essential for reducing environmental impact (Cheng et al., 2021).
- Development in biodiesel by-products treatment: Tartronic acid is also involved in kinetic studies for the electrochemical conversion of glycerol, a by-product of biodiesel production, highlighting its role in renewable energy and waste valorization (Pérès et al., 2020).
- Base-free oxidation reactions: It aids in the development of base-free conditions for glycerol to glyceraldehyde oxidation reactions over platinum-based catalysts, offering advancements in catalysis and organic synthesis processes (Capron et al., 2019).
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
G DuVal et al.
Biochemistry, 24(8), 2067-2072 (1985-04-09)
Some kinetic characteristics of immobilized native mitochondrial malate dehydrogenase dimers and immobilized protomers, prepared by direct immobilization under conditions yielding complete dissociation without substantial unfolding, were compared to those of native soluble enzyme. Enzyme was covalently immobilized to derivatized porous
J K Hiltunen et al.
Biochimica et biophysica acta, 678(1), 115-121 (1981-11-18)
The mechanism of depletion of tricarboxylic acid cycle intermediates by isolated rat heart mitochondria was studied using hydroxymalonate (an inhibitor of malic enzymes) and mercaptopicolinate (an inhibitor of phosphoenolpyruvate carboxykinase) as tools. Hydroxymalonate inhibited the respiration rate of isolated mitochondria
Christopher D Evans et al.
The Journal of chemical physics, 152(13), 134705-134705 (2020-04-10)
The oxidation of glycerol under alkaline conditions in the presence of a heterogeneous catalyst can be tailored to the formation of lactic acid, an important commodity chemical. Despite recent advances in this area, the mechanism for its formation is still
A dynamic kinetic asymmetric transformation in the alpha-hydroxylation of racemic malonates and its application to biologically active molecules.
Dhande Sudhakar Reddy et al.
Angewandte Chemie (International ed. in English), 48(4), 803-806 (2008-12-23)
Formation of 2-keto-D-gluconic acid, 5-keto-D-gluconic acid, and tartronic acid by Acetobacter species.
D KULKA et al.
Nature, 167(4257), 905-906 (1951-06-02)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service