All Photos(1)
About This Item
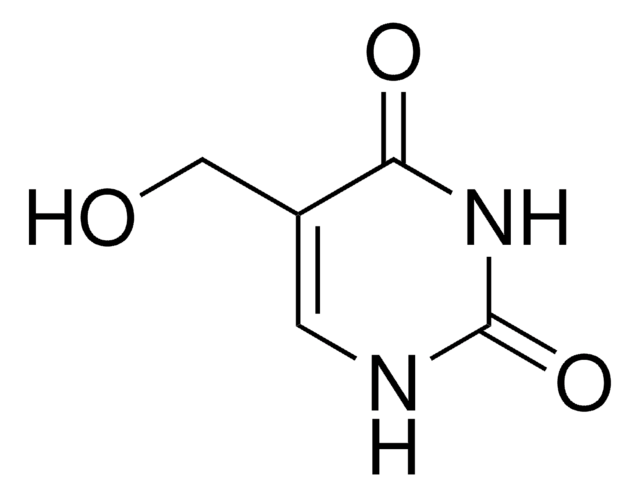
Empirical Formula (Hill Notation):
C5H4N2O3
CAS Number:
Molecular Weight:
140.10
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
powder
mp
>300 °C (dec.) (lit.)
functional group
aldehyde
SMILES string
O=CC1=CNC(=O)NC1=O
InChI
1S/C5H4N2O3/c8-2-3-1-6-5(10)7-4(3)9/h1-2H,(H2,6,7,9,10)
InChI key
OHAMXGZMZZWRCA-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
5-Formyluracil may be used for the preparation of covalently linked base with 5-aminocytosine pair via Schiff base formation.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Q M Zhang et al.
International journal of radiation biology, 79(5), 341-349 (2003-08-29)
5-Formyluracil (5-foU) is a potentially mutagenic lesion of thymine produced in DNA by ionizing radiation and various chemical oxidants. The present authors reported previously that MutM, Nth and Nei in Escherichia coli removed 5-foU from DNA. The present study identified
Gustavo Portalone et al.
Acta crystallographica. Section C, Crystal structure communications, 63(Pt 11), o650-o654 (2007-11-09)
The asymmetric unit of the amino-oxo tautomer of 5-formyluracil (systematic name: 2,4-dioxo-1,2,3,4-tetrahydropyrimidine-5-carbaldehyde), C(5)H(4)N(2)O(3), comprises one planar amino-oxo tautomer, as every atom in the structure lies on a crystallographic mirror plane. At variance with all the previously reported small-molecule crystal structures
Monica Baldini et al.
Inorganic chemistry, 42(6), 2049-2055 (2003-03-18)
Two new 5-formyluracil thiosemicarbazone (H(3)ut) derivatives, Me-H(3)ut (1) and Me(2)-H(3)ut (2), were synthesized by reacting thiosemicarbazides, mono- and dimethylated on the aminic nitrogen, with 5-formyluracil and were subsequently characterized. These ligands, treated with copper chloride and nitrate, afforded three complexes:
Chikara Dohno et al.
Journal of the American Chemical Society, 127(47), 16681-16684 (2005-11-25)
We here present a novel covalently linked base pair via Schiff base formation between 5-formyluracil (fU) and 5-aminocytosine (AmC). Formation of the Schiff base linkage proceeds reversibly and does not require any additives. The cross-linked DNA is very stable under
E J Privat et al.
Mutation research, 354(2), 151-156 (1996-07-22)
5-Formyluracil is a mutagenic base formed in DNA by oxidation of the thymine methyl group. Whereas the thymine methyl group is electron donating, the formyl group is electron withdrawing, predicting increased ionization of the N-3 imino proton under physiological conditions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service