278998
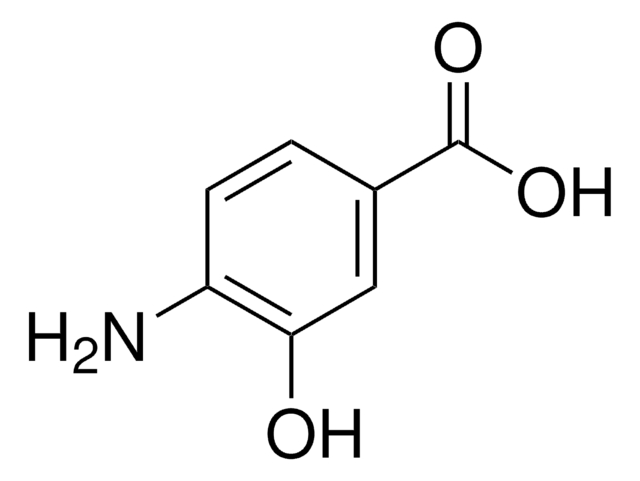
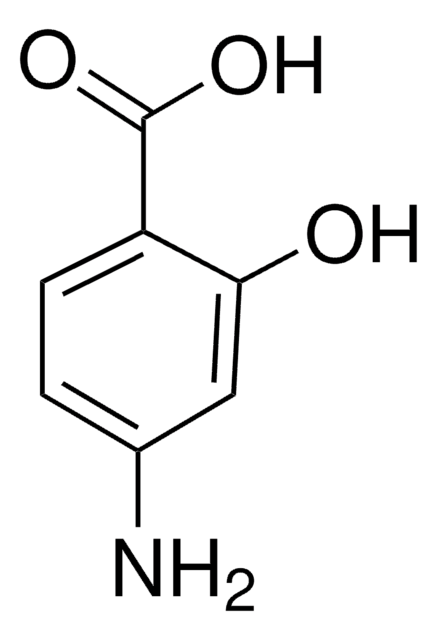
2-Amino-5-hydroxybenzoic acid
98%
Synonym(s):
5-Hydroxyanthranilic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
HOC6H3(NH2)CO2H
CAS Number:
Molecular Weight:
153.14
Beilstein:
2803663
MDL number:
UNSPSC Code:
12352106
eCl@ss:
32160406
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
solid
reaction suitability
reaction type: solution phase peptide synthesis
mp
247 °C (dec.) (lit.)
application(s)
peptide synthesis
SMILES string
Nc1ccc(O)cc1C(O)=O
InChI
1S/C7H7NO3/c8-6-2-1-4(9)3-5(6)7(10)11/h1-3,9H,8H2,(H,10,11)
InChI key
HYNQTSZBTIOFKH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
[Diabeton in the treatment of stage I nephroangiopathy in insulin-dependent diabetes mellitus].
E P Kashirina et al.
Sovetskaia meditsina, (1)(1), 68-69 (1991-01-01)
T A Buĭdina et al.
Sovetskaia meditsina, (7)(7), 14-16 (1991-01-01)
Newly detected noninsulin--dependent diabetes mellitus is shown to have changes in phospholipid structure of erythrocytic membranes: a fall in total phospholipids, sphingomyelin, phosphatidylcholine, phosphatidic acid, a trend to increasing lysophosphatidylcholine. The shifts go in parallel with lipid peroxidation activation. Carbohydrate
V K Velikov et al.
Terapevticheskii arkhiv, 63(10), 44-49 (1991-01-01)
Hemocoagulation was examined in 477 diabetes mellitus patients. Whatever the disease type, severity, duration and the intensity of microvascular complications, diabetes mellitus was discovered to be marked by the development of chronic intravascular blood microcoagulation associated with primary hyperactivation of
Deze Kong et al.
Science advances, 6(44) (2020-11-01)
Chalcone synthase (CHS) canonically catalyzes carbon-carbon bond formation through iterative decarboxylative Claisen condensation. Here, we characterize a previously unidentified biosynthetic capability of SlCHS to catalyze nitrogen-carbon bond formation, leading to the production of a hydroxycinnamic acid amide (HCAA) compound. By
Alexander von Tesmar et al.
Cell chemical biology, 24(10), 1216-1227 (2017-09-12)
In vitro reconstitution and biochemical analysis of natural product biosynthetic pathways remains a challenging endeavor, especially if megaenzymes of the nonribosomal peptide synthetase (NRPS) type are involved. In theory, all biosynthetic steps may be deciphered using mass spectrometry (MS)-based analyses of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service