About This Item
Recommended Products
form
powder
Quality Level
originator
Sanofi Aventis
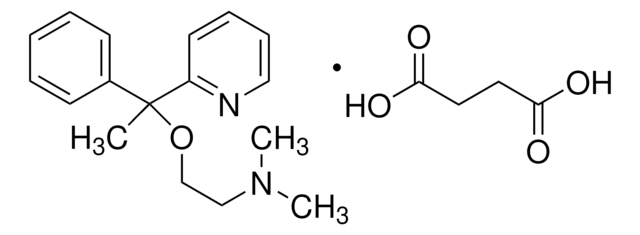
SMILES string
OC(=O)CCC(O)=O.CN(C)CCOC(C)(c1ccccc1)c2ccccn2
InChI
1S/C17H22N2O.C4H6O4/c1-17(20-14-13-19(2)3,15-9-5-4-6-10-15)16-11-7-8-12-18-16;5-3(6)1-2-4(7)8/h4-12H,13-14H2,1-3H3;1-2H2,(H,5,6)(H,7,8)
InChI key
KBAUFVUYFNWQFM-UHFFFAOYSA-N
Gene Information
human ... HRH1(3269)
Looking for similar products? Visit Product Comparison Guide
General description
Application
Biochem/physiol Actions
Doxylamine suppresses histamine at the H1 receptor. It is associated with the short term management of insomnia and temporary relief of common cold symptoms. On the other hand, doxylamine intoxication is linked with rhabdomyolysis and secondary acute renal failure.
Studies in mice show that doxylamine induces liver microsomal cytochrome P450 and other enzymes involved in thyroxine (T4) metabolism. In combination with pyridoxine hydrochloride, this drug is used to treat morning sickness.
Features and Benefits
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service