C8919
Bovine Collagen Type I
from bovine skin, liquid, 1 mg/mL, suitable for cell culture
About This Item
Recommended Products
Product Name
Collagen from calf skin, Bornstein and Traub Type I, (0.1% solution in 0.1 M acetic acid), aseptically processed, BioReagent, suitable for cell culture
biological source
bovine (calf) skin
Quality Level
sterility
aseptically processed
product line
BioReagent
form
solution (0.1% solution in 0.1 M acetic acid)
packaging
pkg of 20 mL
concentration
(0.1% solution in 0.1 M acetic acid)
technique(s)
cell culture | mammalian: suitable
surface coverage
6‑10 μg/cm2
Binding Specificity
Peptide Source: Collagen
Peptide Source: Elastin
Peptide Source: Fibronectin
shipped in
wet ice
storage temp.
2-8°C
Gene Information
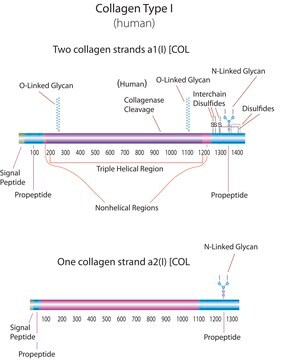
bovine ... COL1A1(282187) , COL2A1(407142)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- for pre-coting glass slides for immunofluorescence studies
- as a cell adhesion factor and modification of poly(vinylidene fluoride-trifluoroethylene) (P(VDF-TrFE)) films for neuron culture
- for coating culture dishes for murine embryonic fibroblasts culture
Biochem/physiol Actions
Components
Preparation Note
Other Notes
Storage Class Code
12 - Non Combustible Liquids
WGK
nwg
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Attachment Factors for 3-Dimensional Cell Culture
The extracellular matrix (ECM) is secreted by cells and surrounds them in tissues.
Cancer stem cell media, spheroid plates and cancer stem cell markers to culture and characterize CSC populations.
Extracellular matrix proteins such as laminin, collagen, and fibronectin can be used as cell attachment substrates in cell culture.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service