A6376
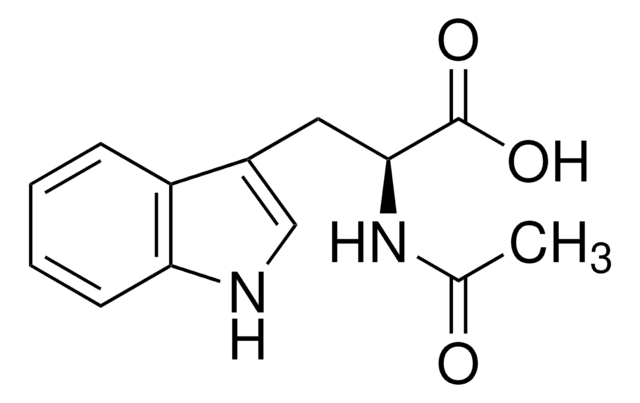
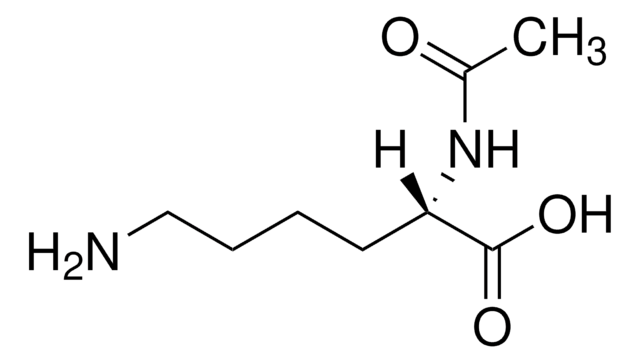
N-Acetyl-L-tryptophan
≥99% (TLC), suitable for ligand binding assays
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
C13H14N2O3
CAS Number:
Molecular Weight:
246.26
EC Number:
MDL number:
UNSPSC Code:
12352209
eCl@ss:
32160406
PubChem Substance ID:
NACRES:
NA.26
Recommended Products
Product Name
N-Acetyl-L-tryptophan,
Assay
≥99% (TLC)
Quality Level
form
powder
technique(s)
ligand binding assay: suitable
color
white to off-white
storage temp.
2-8°C
SMILES string
CC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O
InChI
1S/C13H14N2O3/c1-8(16)15-12(13(17)18)6-9-7-14-11-5-3-2-4-10(9)11/h2-5,7,12,14H,6H2,1H3,(H,15,16)(H,17,18)/t12-/m0/s1
InChI key
DZTHIGRZJZPRDV-LBPRGKRZSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Biochem/physiol Actions
N-Acetyl-L-tryptophan (NAT, Ac-Trp-OH) is used a substance P NK1 tachykinin receptor antagonist. N-Acetyl-L-tryptophan is also used as a competitive inhibitor to identify, differentiate and characterized tryptophanase(s).
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
James J Donkin et al.
Journal of neurotrauma, 28(2), 217-224 (2010-12-24)
Previous studies have demonstrated that the compound N-acetyl-L-tryptophan (NAT) reduces brain edema and improves functional outcome following traumatic brain injury (TBI). In this study we examined whether this effect was mediated via the neurokinin-1 receptor, and whether there was an
New tryptophanase inhibitors: towards prevention of bacterial biofilm formation.
Scherzer R, Gdalevsky GY, et al.
Journal of Enzyme Inhibition, 24, 350-355 (2009)
Kate M Lewis et al.
Anti-cancer drugs, 24(4), 344-354 (2013-02-15)
Emend, an NK1 antagonist, and dexamethasone are used to treat complications associated with metastatic brain tumours and their treatment. It has been suggested that these agents exert anticancer effects apart from their current use. The effects of the NK1 antagonists
Toshinori Suzuki et al.
Free radical biology & medicine, 37(5), 671-681 (2004-08-04)
Proteins are targets of reactive nitrogen species such as peroxynitrite and nitrogen dioxide. Among the various amino acids in proteins, tryptophan residues are especially susceptible to attack by reactive nitrogen species. We carried out experiments on the reactions of peroxynitrite
Renée J Turner et al.
Brain research, 1393, 84-90 (2011-04-07)
Previous studies have suggested that substance P (SP) plays a critical role in the development of brain oedema and functional deficits following traumatic brain injury and that SP receptor antagonism may improve outcome. No studies have described such a role
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service