173401
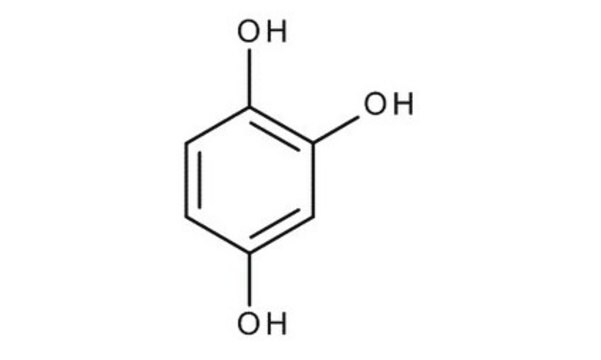
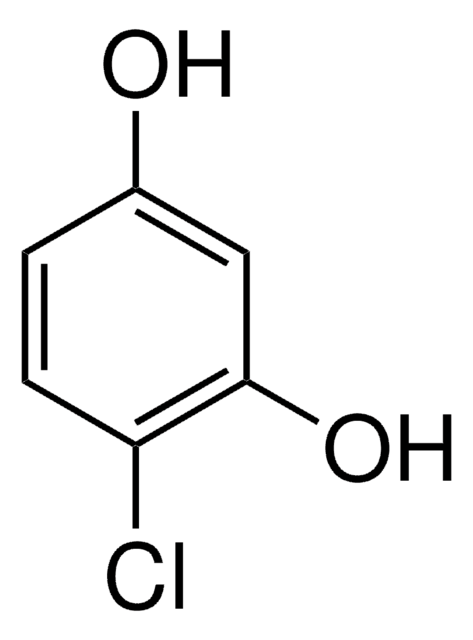
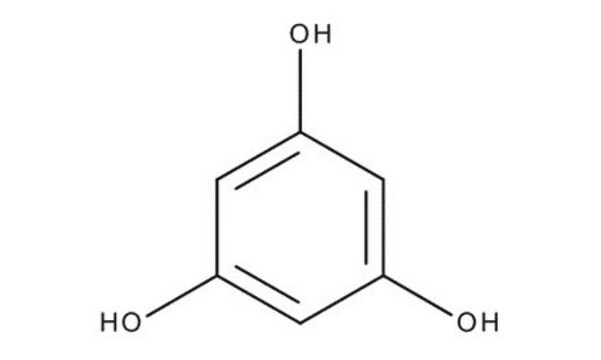
1,2,4-Benzenetriol
ReagentPlus®, 99%
Synonym(s):
Hydroxyhydroquinone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
C6H3(OH)3
CAS Number:
Molecular Weight:
126.11
EC Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
Quality Level
product line
ReagentPlus®
Assay
99%
form
solid
mp
140 °C (subl.) (lit.)
SMILES string
Oc1ccc(O)c(O)c1
InChI
1S/C6H6O3/c7-4-1-2-5(8)6(9)3-4/h1-3,7-9H
InChI key
GGNQRNBDZQJCCN-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
1,2,4-Benzenetriol is also known as hydroxyhydroquinone. It is an intermediary metabolite of benzene that is present in roasted coffee beans. It is mutagenic and it causes cleaving of DNA single strands by the generation of reactive oxygen species.
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Not finding the right product?
Try our Product Selector Tool.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Generation of hydrogen peroxide from hydroxyhydroquinone and its inhibition by superoxide dismutase
Hiramoto K, et al.
Journal of Oleo Science, 50(1), 21-28 (2001)
Karolina Nordin et al.
Applied and environmental microbiology, 71(11), 6538-6544 (2005-11-05)
Arthrobacter chlorophenolicus A6, a previously described 4-chlorophenol-degrading strain, was found to degrade 4-chlorophenol via hydroxyquinol, which is a novel route for aerobic microbial degradation of this compound. In addition, 10 open reading frames exhibiting sequence similarity to genes encoding enzymes
Rolis Chien-Wei Hou et al.
Journal of biomedical science, 13(1), 89-99 (2005-11-26)
Hydroxyhydroquinone or 1,2,4-benzenetriol (BT) detected in the beverages has a structure that coincides with the water-soluble form of a sesame lignan, sesamol. We previously showed that sesame antioxidants had neuroprotective abilities due to their antioxidant properties and/or inducible nitric oxide
Atsushi Suzuki et al.
FEBS letters, 580(9), 2317-2322 (2006-04-04)
Chlorogenic acid, a polyphenol found in coffee, has antihypertensive actions, but epidemiologic data on the effects of coffee on blood pressure are controversial. Specific coffee components that inhibit the hypotensive effect of chlorogenic acid and the physiologic mechanisms underlying the
Luoping Zhang et al.
Environmental and molecular mutagenesis, 45(4), 388-396 (2005-01-22)
The loss and gain of whole chromosomes (aneuploidy) is common in the development of leukemia and other cancers. In acute myeloid leukemia, the loss (monosomy) of chromosomes 5 and 7 and the gain (trisomy) of chromosome 8 are common clonal
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service