All Photos(1)
About This Item
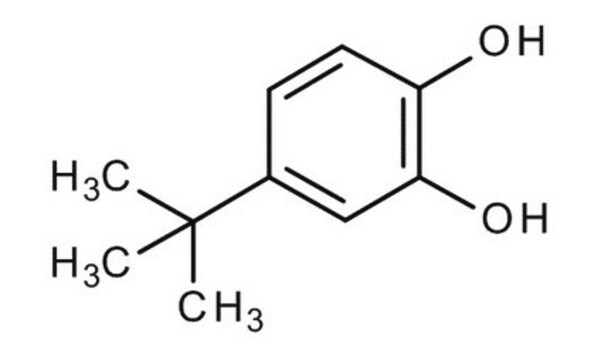
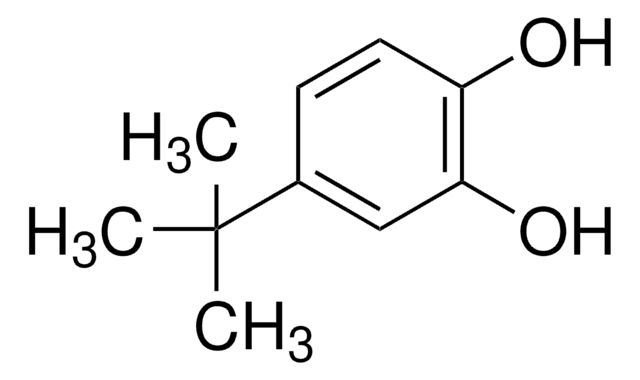
Linear Formula:
(CH3)3CC6H3-1,2-(OH)2
CAS Number:
Molecular Weight:
166.22
Beilstein:
2043335
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
Recommended Products
Assay
97%
bp
285 °C (lit.)
mp
52-55 °C (lit.)
SMILES string
CC(C)(C)c1ccc(O)c(O)c1
InChI
1S/C10H14O2/c1-10(2,3)7-4-5-8(11)9(12)6-7/h4-6,11-12H,1-3H3
InChI key
XESZUVZBAMCAEJ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
4-tert-Butylcatechol undergoes electrochemical trimerization via anodic oxidation and mechanism has been studied by cyclic voltammetry and controlled-potential coulometry. It inhibits the activity of tyrosinase at concentrations higher than 1×10−3M. It undergoes oxidation with laccase to yield quinones which on reaction with dienes and oxidation afford naphthoquinones.
Application
4-tert-Butylcatechol was used in the synthesis of tungsten oxide nanoparticles by nonaqueous sol-gel process.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Corr. 1B - Skin Sens. 1
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Y Usami et al.
Journal of toxicology and environmental health, 6(3), 559-567 (1980-05-01)
4-tert-Butylcatechol (TBC) is an antioxidant widely used in industry and a potent depigmenting agent to the skin of the workers. In this study, tyrosinase was extracted from tissue-cultured human melanoma cells and purified by polyacrylamide gel electrophoresis. T1 and T2
Ligand and solvent effects in the nonaqueous synthesis of highly ordered anisotropic tungsten oxide nanostructures.
Polleux J, et al.
Journal of Materials Chemistry, 16(40), 3969-3975 (2006)
Tetrahedron Letters, 48, 2983-2983 (2007)
Mechanistic study of electrochemical oxidation of 4-tert-butylcatechol: A facile electrochemical method for the synthesis of new trimer of 4-tert-butylcatechol.
Nematollahi D, et al.
Electrochimica Acta, 49(15), 2495-2502 (2004)
J N Rodriguez-López et al.
Analytical biochemistry, 202(2), 356-360 (1992-05-01)
A procedure for calibrating a Clark-type oxygen electrode is described. This method is based on the oxidation of 4-tert-butylcatechol (TBC) by O2 catalyzed by tyrosinase, to yield 4-tert-butyl-o-benzoquinone (TBCQ). This reaction consumes known amounts of oxygen in accordance with the
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service