670871
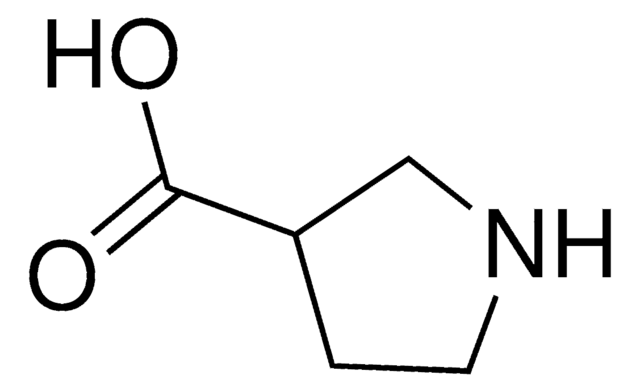
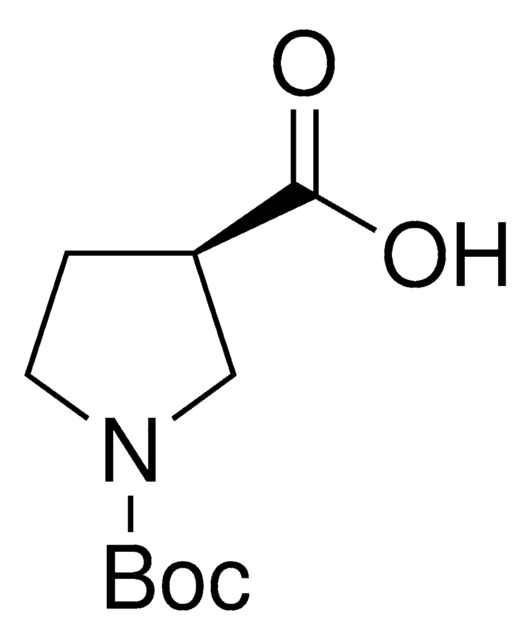
(S)-(+)-Pyrrolidine-3-carboxylic acid
≥98.0% (NT)
Synonym(s):
(S)-β-Proline
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H9NO2
CAS Number:
Molecular Weight:
115.13
Beilstein:
3537293
MDL number:
UNSPSC Code:
12352106
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
≥98.0% (NT)
form
solid
optical activity
[α]/D 20.0±2.0°, c = 2 in H2O
reaction suitability
reaction type: solution phase peptide synthesis
application(s)
peptide synthesis
storage temp.
2-8°C
SMILES string
OC(=O)[C@H]1CCNC1
InChI
1S/C5H9NO2/c7-5(8)4-1-2-6-3-4/h4,6H,1-3H2,(H,7,8)/t4-/m0/s1
InChI key
JAEIBKXSIXOLOL-BYPYZUCNSA-N
Related Categories
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
David Steer et al.
Biochemistry, 41(35), 10819-10826 (2002-08-28)
The enzyme EC 3.4.24.15 (EP 24.15) is a zinc metalloendopeptidase whose precise function in vivo remains unknown but is thought to participate in the regulated metabolism of a number of specific neuropeptides. The lack of stable and selective inhibitors has
Wesley R R Harker et al.
Organic & biomolecular chemistry, 10(7), 1406-1410 (2012-01-05)
α-Alkyl β-amino esters are available in high diastereoselectivity through a silicon-free Claisen enolate [3,3]-sigmatropic rearrangement of enamide esters. Optimisation studies have probed the crucial role of the initial enolisation and the nature of the enamide N-centre. The demonstration of chirality
Haile Zhang et al.
Journal of the American Chemical Society, 130(3), 875-886 (2008-01-01)
The development of enantioselective anti-selective Mannich-type reactions of aldehydes and ketones with imines catalyzed by 3-pyrrolidinecarboxylic acid and related pyrrolidine derivatives is reported in detail. Both (3R,5R)-5-methyl-3-pyrrolidinecarboxylic acid and (R)-3-pyrrolidinecarboxylic acid efficiently catalyzed the reactions of aldehydes with alpha-imino esters
Cody Timmons et al.
The Journal of organic chemistry, 70(19), 7634-7639 (2005-09-10)
[reaction: see text] A new halo-Mannich-type reaction is reported using cyclopropyl carbonyl-derived enolates and sulfonyl-protected imines. Chiral oxazolidinones auxiliaries were found to be effective for completely controlling the stereochemistry of the products. Variations in the oxazolidinone, protecting group, and imine
Souvik Banerjee et al.
The Journal of organic chemistry, 77(23), 10925-10930 (2012-11-07)
A straightforward stereoselective and enantiodivergent cyclization strategy for the construction of γ-lactams is described. The cyclization strategy makes use of chiral malonic esters prepared from enantiomerically enriched monoesters of disubstituted malonic acid. The cyclization occurs with the selective displacement of
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service