569437
tert-Butylsulfinyl chloride
97%
Synonym(s):
1,1-Dimethylethanesulfinyl chloride, 2-Methyl-2-propanesulfinyl chloride, 2-Methylpropane-2-sulfinic chloride, tert-Butylthionyl chloride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
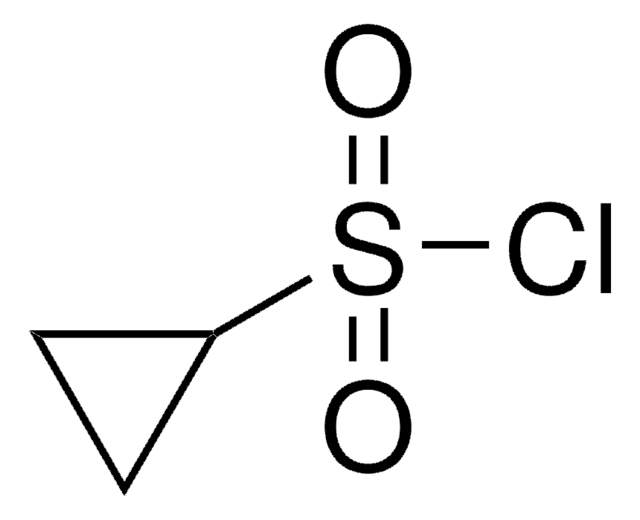
(CH3)3CS(O)Cl
CAS Number:
Molecular Weight:
140.63
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
refractive index
n20/D 1.4830 (lit.)
bp
53-54 °C/19 mmHg (lit.)
density
1.135 g/mL at 25 °C (lit.)
SMILES string
CC(C)(C)S(Cl)=O
InChI
1S/C4H9ClOS/c1-4(2,3)7(5)6/h1-3H3
InChI key
ZLJKQOWJEZSNBE-UHFFFAOYSA-N
General description
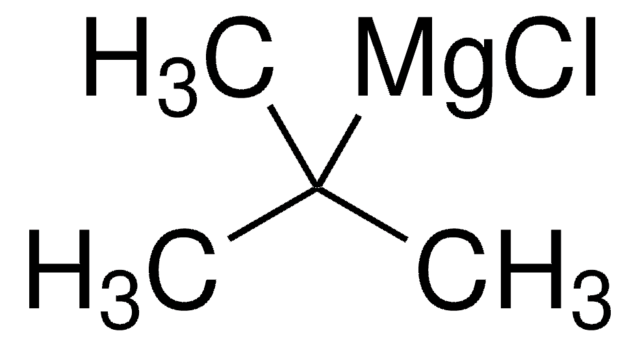
tert-Butylsulfinyl chloride can be synthesized by using tert-butylmagnesium chloride as the starting material via a multi-step synthesis process.
Application
tert-butylsulfinyl chloride can react with primary and secondary amines to form tert-butylsulfinamides, which on oxidation forms base stable tert-butylsulfonamide. It can also react with 1-thiosugars to form the corresponding carbohydrate thionolactones.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Flam. Liq. 3 - Skin Corr. 1B
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
125.1 °F - closed cup
Flash Point(C)
51.7 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
"One-pot synthesis of carbohydrate thionolactones from 1-thiosugars"
Wilkinson.LB and Fairbanks.JA
Tetrahedron Letters, 49(33), 4941-4943 (2008)
"Tert-butylsulfonyl (Bus), a new protecting group for amines"
Sun P, et al.
The Journal of Organic Chemistry, 62(24), 8604-8608 (1997)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service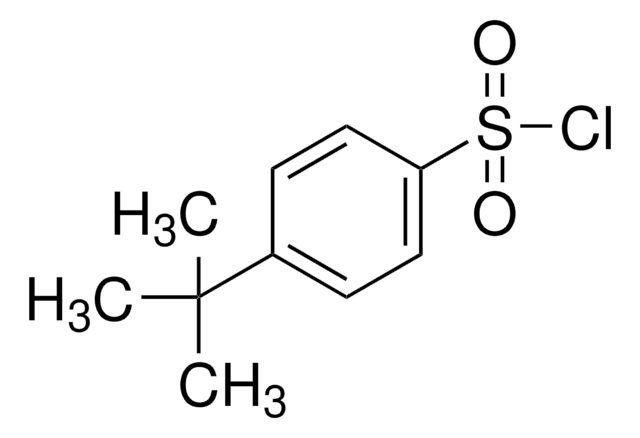




![(R)-N-[(1R,2R)-2-(3-(3,5-Bis(trifluoromethyl)phenyl)ureido)cyclohexyl]-tert-butyl-sulfinamide 96%](/deepweb/assets/sigmaaldrich/product/structures/389/070/18847164-c6a7-4b4e-abcb-2dbc22493a2d/640/18847164-c6a7-4b4e-abcb-2dbc22493a2d.png)