345474
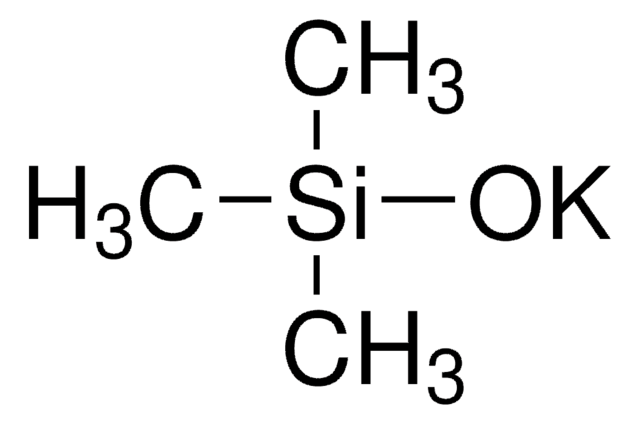
Lithium trimethylsilanolate
95%
Synonym(s):
Trimethylsilanol lithium salt
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
(CH3)3SiOLi
CAS Number:
Molecular Weight:
96.13
Beilstein:
3686900
MDL number:
UNSPSC Code:
12352000
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
95%
form
solid
SMILES string
[Li+].C[Si](C)(C)[O-]
InChI
1S/C3H9OSi.Li/c1-5(2,3)4;/h1-3H3;/q-1;+1
InChI key
OXOZHAWWRPCVGL-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- Enhanced process and composition control for atomic layer deposition with lithium trimethylsilanolate: This study reports on the novel lithium precursor, lithium trimethylsilanolate (LiTMSO), for atomic layer deposition, comparing it with other lithium precursors for improved process and composition control (Ruud et al., 2017).
- Trimethylsiloxy based metal complexes as electrolyte additives for high voltage application in lithium ion cells: The paper discusses the synthesis of lithium trimethylsilanolate and its use as an additive to enhance the performance of high voltage lithium ion cells (Imholt et al., 2017).
- Application of trimethylsilanolate alkali salts in organic synthesis: This review covers various applications of lithium trimethylsilanolate in organic synthesis, highlighting its role in different reactions (Bürglová and Hlaváč, 2018).
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
D M Hui et al.
Clinica chimica acta; international journal of clinical chemistry, 302(1-2), 171-188 (2000-11-14)
We developed a new assay method for fluoride anion (F(-)) a specific metabolite of sarin. Trimethyifluorosilane (TMFS) was derivatized from F(-) with trimethylsilanol, and TMFS was detected with a GC-flame ionization detector (FID) and capillary column system. The linear range
A Isquith et al.
Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 26(3), 263-266 (1988-03-01)
Six organosilicon compounds which had been found to have clastogenic activity in an in vitro battery of genotoxicity assays were evaluated in rat bone marrow cytogenetic assays for assessing clastogenicity in an in vivo system. None of the six compounds
Igor S Ignatyev et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 60(5), 1169-1178 (2004-04-16)
The assignment of the SiOH group vibrations of trimethylsilanol, which is still controversial, is proposed. This assignment is based on theoretical B3LYP force field scaled using the constants of the (CH3)3Si group optimized to fit experimental vibrational frequencies of (CH3)3SiF
Fabricio A Hansel et al.
Rapid communications in mass spectrometry : RCM, 25(13), 1893-1898 (2011-06-04)
A methodology is presented for the determination of dihydroxy fatty acids preserved in the 'bound' phase of organic residues preserved in archaeological potsherds. The method comprises saponification, esterification, silica gel column chromatographic fractionation, and analysis by gas chromatography/mass spectrometry. The
Diane M Coe et al.
Organic & biomolecular chemistry, 1(7), 1106-1111 (2003-08-21)
A convenient and efficient method for the cleavage of 1,3-oxazolidin-5-ones and 1,3-oxazolidin-2-ones utilising potassium trimethylsilanolate in tetrahydrofuran is described. The benzyloxycarbonyl-protecting group is readily removed under the reaction conditions, whereas the N-benzoyl group is stable. A synthesis of (R)-salmeterol exploiting
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service