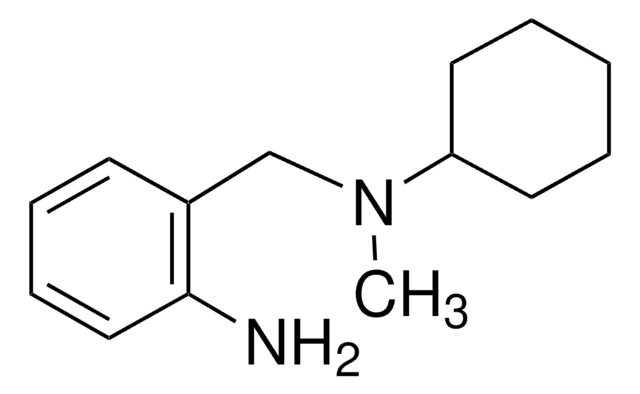
294942
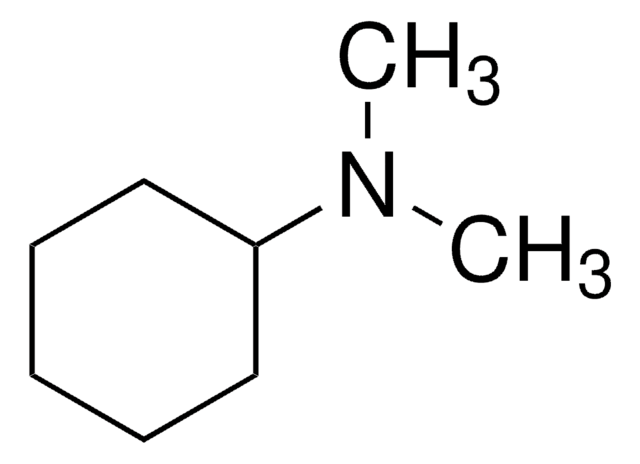
N,N-Dicyclohexylmethylamine
97%
Synonym(s):
N-Methyldicyclohexylamine
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
(C6H11)2NCH3
CAS Number:
Molecular Weight:
195.34
Beilstein:
2074991
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
refractive index
n20/D 1.49 (lit.)
bp
265 °C (lit.)
density
0.912 g/mL at 25 °C (lit.)
functional group
amine
SMILES string
CN(C1CCCCC1)C2CCCCC2
InChI
1S/C13H25N/c1-14(12-8-4-2-5-9-12)13-10-6-3-7-11-13/h12-13H,2-11H2,1H3
InChI key
GSCCALZHGUWNJW-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
N,N-Dicyclohexylmethylamine has been employed as:
- catalyst during O-phenylation of tertiary alcohol with organobismuth(V) compounds
- base during Pd-catalyzed 5-endo-trig cyclization of 1-(o-bromophenyl)-2-methylprop-2-en-1-ol
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 2
Flash Point(F)
230.0 °F - closed cup
Flash Point(C)
110 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Sriram Kumar et al.
Nature communications, 11(1), 2960-2960 (2020-06-13)
Nucleic acid-based materials enable sub-nanometer precision in self-assembly for fields including biophysics, diagnostics, therapeutics, photonics, and nanofabrication. However, structural DNA nanotechnology has been limited to substantially hydrated media. Transfer to organic solvents commonly used in polymer and peptide synthesis results
Copper (II)-catalyzed O-phenylation of alcohols with organobismuth (V) reagents.
Sakurai N, et al.
ARKIVOC (Gainesville, FL, United States), 7, 254-264 (2007)
Heck-type reactions of allylic alcohols: Part IV:(2-Substituted)-1-indanones via 5-endo-trig cyclizations.
Zawisza AM, et al.
J. Mol. Catal. A: Chem., 283(1), 140-145 (2008)
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 294942-1KG | 4061826603741 |
| 294942-250G | 4061826603819 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service