P4932
Polymyxin B sulfate salt
powder, non-animal origin, suitable for cell culture, BioReagent
Synonym(s):
Polymixin, Polymixin B, Polymixin B sulfate
About This Item
Recommended Products
Product Name
Polymyxin B sulfate salt, powder, BioReagent, suitable for cell culture
product line
BioReagent
Quality Level
form
powder
potency
≥6,000 USP units per mg
technique(s)
cell culture | mammalian: suitable
antibiotic activity spectrum
Gram-negative bacteria
Mode of action
cell membrane | interferes
storage temp.
2-8°C
SMILES string
O=C(C(NC(C(CCN)NC(CCCCC(C)CC)=O)=O)C(C)O)NC(CCN)C(NC(CCNC(C(NC(C(NC(C(CCN)N1)=O)CCN)=O)C(O)C)=O)C(NC(CCN)C(NC(CC2=CC=CC=C2)C(NC(CC(C)C)C1=O)=O)=O)=O)=O.O=S(O)(O)=O
InChI
1S/C48H82N16O13.H2O4S/c1-27(2)24-37-47(76)59-32(11-19-52)41(70)56-31(10-18-51)43(72)61-35(14-22-65)39(68)54-21-13-34(45(74)57-33(12-20-53)44(73)64-38(48(77)63-37)25-28-6-4-3-5-7-28)60-42(71)30(9-17-50)58-46(75)36(15-23-66)62-40(69)29(8-16-49)55-26-67;1-5(2,3)4/h3-7,26-27,29-38,65-66H,8-25,49-53H2,1-2H3,(H,54,68)(H,55,67)(H,56,70)(H,57,74)(H,58,75)(H,59,76)(H,60,71)(H,61,72)(H,62,69)(H,63,77)(H,64,73);(H2,1,2,3,4)
InChI key
HNDFYNOVSOOGDU-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Chemical structure: peptide
Application
- for the treatment of BAC-1.2F5 murine macrophages as an LPS (lipopolysaccharide) inhibitor
- as positive and negative control during microbicidal concentration (MCC) assay
Biochem/physiol Actions
Antimicrobial spectrum: Has bactericidal action on most gram-negative bacilli††, including E. Coli and on most fungi and gram-positive bacteria.
Caution
Preparation Note
Other Notes
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service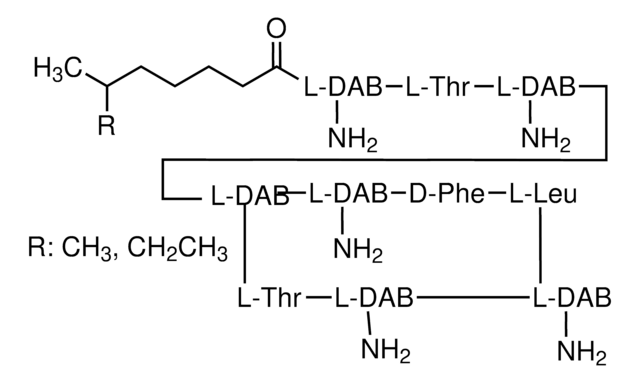


![Benzo[a]pyrene ≥96% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/253/820/be96d879-1811-46c0-8f11-612019691c2d/640/be96d879-1811-46c0-8f11-612019691c2d.png)