8.51006
HBTU
≥99.0% (HPLC), for peptide synthesis, Novabiochem®
Synonym(s):
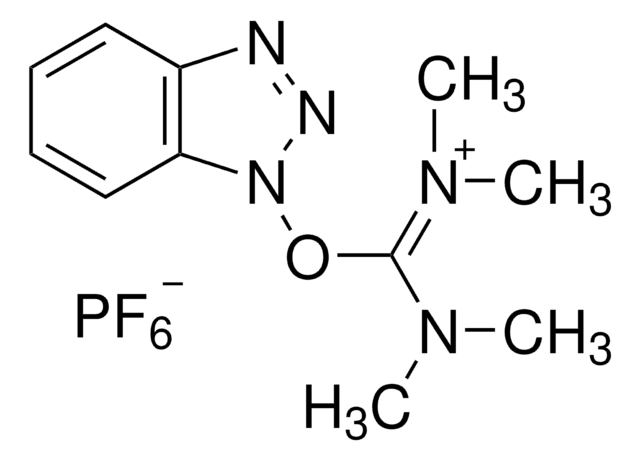
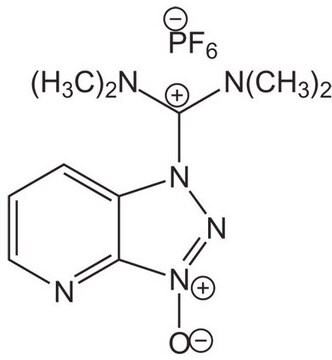
HBTU, 2-(1H-Benzotriazole-1-yl)-1,1,3,3-tetramethylaminium hexafluorophosphate
About This Item
Recommended Products
product name
HBTU, 2-(1H-Benzotriazole-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate Novabiochem®
Quality Level
product line
Novabiochem®
Assay
≥99.0% (HPLC)
form
powder
potency
>2000 mg/kg LD50, oral (Rat)
reaction suitability
reaction type: Coupling Reactions
manufacturer/tradename
Novabiochem®
pH
4.1 (1.6 g/L in H2O)
mp
250 °C
solubility
1.6 g/L
application(s)
peptide synthesis
storage temp.
2-8°C
InChI
1S/C11H16N5O/c1-14(2)11(15(3)4)17-16-10-8-6-5-7-9(10)12-13-16/h5-8H,1-4H3/q+1
InChI key
CLZISMQKJZCZDN-UHFFFAOYSA-N
General description
Associated Protocols and Technical Articles
Guide to Selection of Coupling Reagents
Literature references
[1] R. Knorr, et al. (1989) Tetrahedron Lett., 30, 1927.
[2] M. S. Bernatowicz, et al. (1989) Tetrahedron Lett., 30, 4645.
[3] D. Ambrosius, et al. (1989) Biol. Chem. Hoppe-Seyler, 370, 217.
[4] C. G. Fields, et al. (1991) Pept. Res., 4, 95.
[5] A. G. Beck-Sickinger, et al. (1991) Pept. Res., 4, 88.
[6] G. E. Reid, et al. (1992) Anal. Biochem., 200, 301.
[7] G. B. Fields, et al. in ′Innovation & Perspectives in Solid Phase Synthesis, 1st International Symposium′, R. Epton (Eds), SPCC UK Ltd., Birmingham, 1990, pp. 241.
[8] P. A. Baybayan, et al. in ′Peptides, Chemistry & Biology, Proc. 12th American Peptide Symposium′, J. A. Smith & J. E. Rivier (Eds), ESCOM, Leiden, 1992, pp. 566.
[9] J. J. Dudash, et al. (1993) Synth. Commun., 23, 349.
Application
- Synthesis of Quinoxaline Derivatives Using HBTU: A study highlighting the use of HBTU as a Lewis acid catalyst for synthesizing quinoxaline derivatives, presenting a mild and green protocol (Popatkar and Meshram, 2020).
- Efficient Conversion of Carboxylic Acids into Benzimidazoles: Details an HBTU-promoted methodology for converting carboxylic acids into benzimidazoles in a one-pot strategy (Barasa and Yoganathan, 2018).
- Synthesis of Malonyl-linked Glycoconjugates: Discusses the use of HBTU in the synthesis of glycoconjugates, comparing its efficiency with other reagents (Nörrlinger et al., 2016).
Linkage
Analysis Note
Appearance of substance (visual): powder
Identity (IR): passes test
Assay (HPLC, area%): ≥ 99.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
Legal Information
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Sens. 1A
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Novabiochem® offers a large number of coupling reagents for in situ activation. In situ activating reagents are easy to use, fast reacting – even with sterically hindered amino acids, and their use is generally free of side reactions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service