All Photos(3)
About This Item
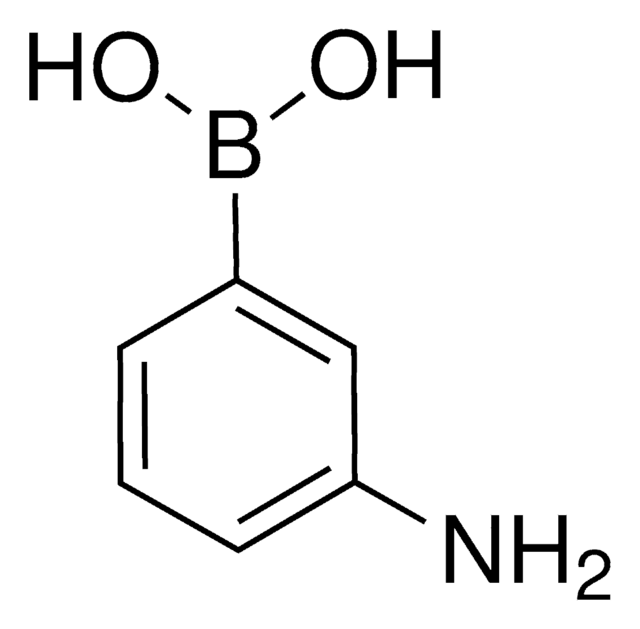
Empirical Formula (Hill Notation):
C13H11BO4
CAS Number:
Molecular Weight:
242.04
MDL number:
UNSPSC Code:
12352106
Recommended Products
Assay
≥95%
form
powder or crystals
reaction suitability
reagent type: catalyst
greener alternative product characteristics
Catalysis
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp
150-152 °C
greener alternative category
storage temp.
2-8°C
General description
We are committed to bringing you Greener Alternative Products, which adhere to one or more of The 12 Principles of Greener Chemistry. This product has been enhanced for catalytic efficiency. Click here for more information.
Application
(2-(Phenoxycarbonyl)phenyl)boronic acid is a boronic acid catalyst from the Hall Group developed for chemoselective activation of oxime N−OH bonds for the preparation of functionalized amide compounds via Beckmann Rearrangement. This catalyst is efficient to transform various diaryl, aryl-alkyl, heteroaryl-alkyl, and dialkyl oximes to amide products under ambient conditions.
related product
Product No.
Description
Pricing
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Xiaobin Mo et al.
Journal of the American Chemical Society, 140(15), 5264-5271 (2018-03-23)
Catalytic activation of hydroxyl functionalities is of great interest for the production of pharmaceuticals and commodity chemicals. Here, 2-alkoxycarbonyl- and 2-phenoxycarbonyl-phenylboronic acid were identified as efficient catalysts for the direct and chemoselective activation of oxime N-OH bonds in the Beckmann
Scope and Mechanism of a True Organocatalytic Beckmann Rearrangement with a Boronic Acid/Perfluoropinacol System under Ambient Conditions.
Mo X, et al.
Journal of the American Chemical Society, 140(15), 5264-5271 (2018)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service

![[Pd(OAc)2]3 reagent grade, 98%](/deepweb/assets/sigmaaldrich/product/structures/508/249/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f/640/99a0ef2c-b77c-4d73-8ed9-0cca05b6b41f.png)
