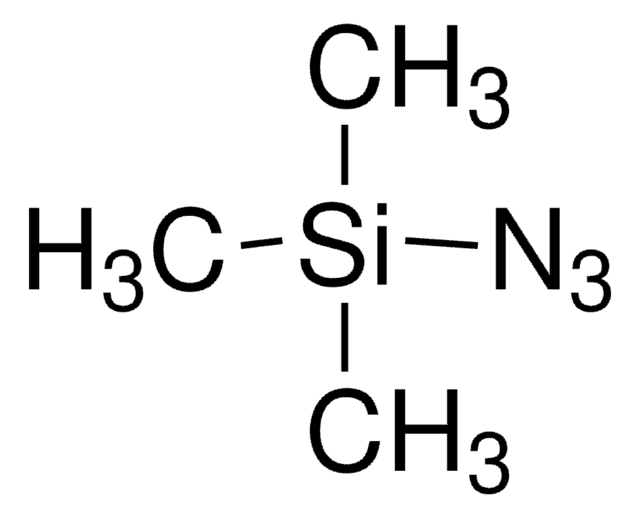740411
Potassium azide
≥99.9% trace metals basis
Synonym(s):
Azidopotassium
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
KN3
CAS Number:
Molecular Weight:
81.12
EC Number:
UNSPSC Code:
12352125
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
≥99.9% trace metals basis
form
solid
mp
~300 °C (decomposition)
functional group
azide
SMILES string
[K+].[N-]=[N+]=[N-]
InChI
1S/K.N3/c;1-3-2/q+1;-1
InChI key
TZLVRPLSVNESQC-UHFFFAOYSA-N
Application
Potassium azide can be used as a reagent to synthesize:
- 3-O-(3-Azidoopropyl)estrone from 3-O-(3-methanesulfonyloxypropoxy)estrone via nucleophilic azidation in the presence of imidazolium catalyst.
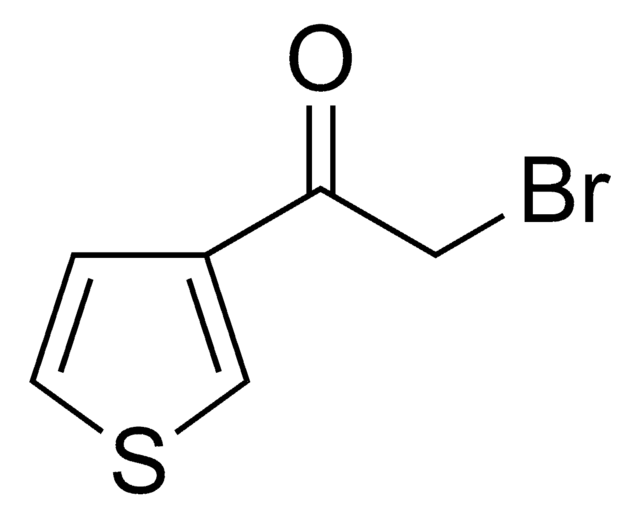
- Azido phenacyl derivatives from α-bromoacetophenone derivatives in the presence of tetrabutylammonium bromide as a catalyst.
- 1-Azido-4-fluorobenzene from potassium (4-fluorophenyl)trifluoroborate in the presence of cupric triflate as a catalyst.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 2 Oral
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
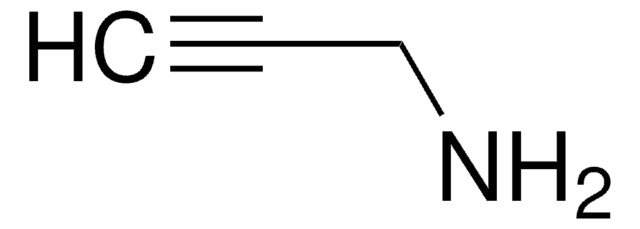
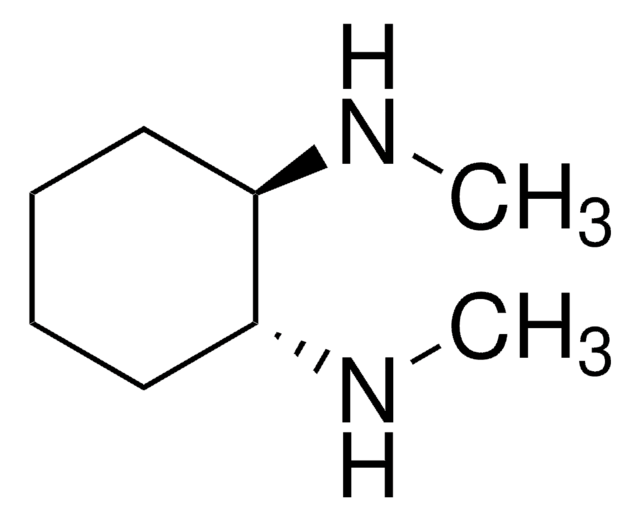
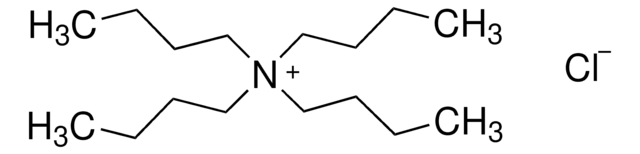
Customers Also Viewed
A M Egorov et al.
Biochimica et biophysica acta, 659(1), 141-149 (1981-05-14)
Modification of 12 arginine residues per molecule of formate dehydrogenase (formate : NAD+ oxidoreductase, EC 1.2.1.2.) from the methylotrophic bacterium, Achromobacter parvulus I, by 2,3-butanedione results in complete inactivation of the enzyme. Inactivation of the enzyme is reversible and proceeds
An efficient method for synthesis of phenacyl derivatives under homogeneous phase transfer catalyst condition in aqueous media
Sayyahi S, et al.
Chinese Chemical Letters = Zhongguo Hua Xue Kuai Bao, 22, 300-302 (2011)
tert-Alcohol-functionalized imidazolium ionic liquid: catalyst for mild nucleophilic substitution reactions at room temperature
Shinde S, et al.
Tetrahedron Letters, 50, 6654-6657 (2009)
A M Liashchuk et al.
Zhurnal mikrobiologii, epidemiologii, i immunobiologii, (4)(4), 65-68 (2006-09-01)
A highly purified TUL4-CBD chimeric protein was obtained by one stage purification method. TUL4-CBD protein consists of TUL4 Francisella tularensis mature peptide sequence, Gly-Ser spacer and cellulose binding domain (CBD) of Anaerocellum thermophilum. The TUL4-CBD protein was shown to induce
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service