All Photos(1)
About This Item
Empirical Formula (Hill Notation):
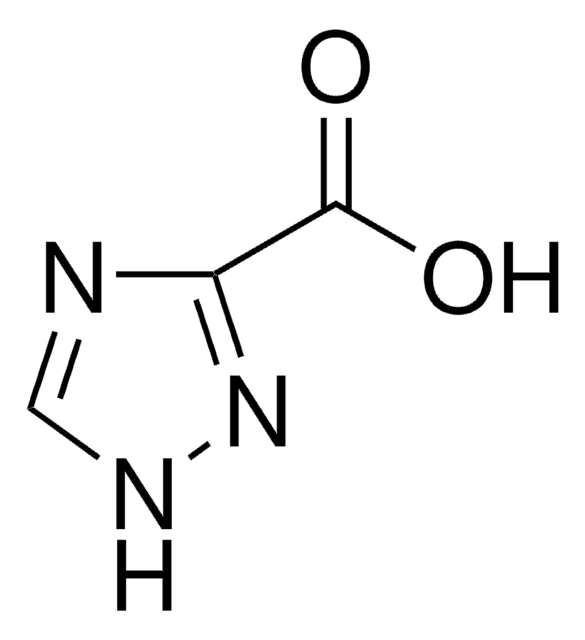
C4H5N3O2
CAS Number:
Molecular Weight:
127.10
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
mp
196-199 °C (lit.)
functional group
ester
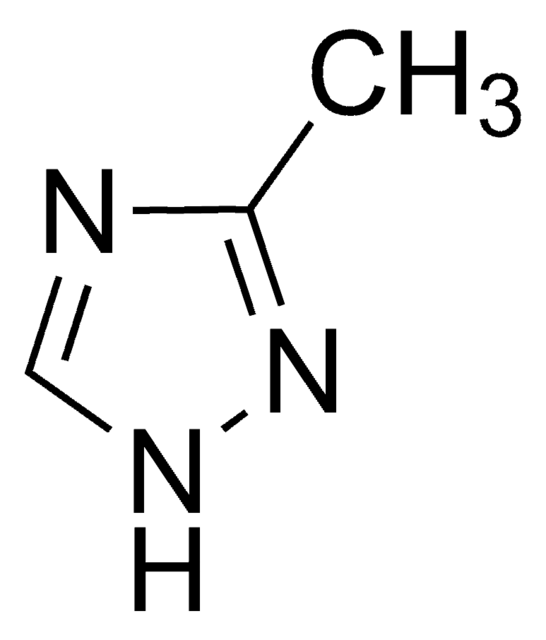
SMILES string
COC(=O)c1nc[nH]n1
InChI
1S/C4H5N3O2/c1-9-4(8)3-5-2-6-7-3/h2H,1H3,(H,5,6,7)
InChI key
QMPFMODFBNEYJH-UHFFFAOYSA-N
General description
Methyl-1H-1,2,4-triazole-3-carboxylate can be synthesized from 5-amino-1,2,4-triazole-3-carboxylic acid via esterification with methanol. It is utilized as precursor for preparing the nucleoside analogue, Ribavirin. The crystal structure of methyl-1H-1,2,4-triazole-3-carboxylate has been analyzed.
Application
Methyl-1H-1,2,4-triazole-3-carboxylate (1,2,4-Triazole-3-methylcarboxylate) may be used in the preparation of 1H-1,2,4-triazole-3-carbohydrazide.
It may also be used in the synthesis of the following nucleoside analogues:
It may also be used in the synthesis of the following nucleoside analogues:
- 1-(2,2-dimethyl-6-trityloxymethyl-4,6a-dihydro-3aH-cyclopenta-[1,3]dioxol-4-yl)-1H-1,2,4-triazole-3-carboxylic acid methyl ester
- methyl 4,6-O-benzylidene-2-(methyl 1H-1,2,4-triazol-1-yl-3-carboxylate)-2-deoxy-<SC>D</SC>-altrohexopyranoside
- 1,5-anhydro-4,6-O-benzylidene-2-(methyl 1H-1,2,4-triazol-1-yl-3-carboxylate)-2-deoxy-<SC>D</SC>-altro-hexitol
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Dinuclear, Tetranuclear and Chain (MnII, CoII) Complexes of Multifunctional Hydrazone Ligands?Structural and Magnetic Studies.
Bettle PJ, et al.
European Journal of Inorganic Chemistry, 2011(32), 5036-5042 (2011)
Arthur Van Aerschot et al.
Antiviral chemistry & chemotherapy, 14(1), 23-30 (2003-06-07)
Current standard therapy for the treatment of chronic infections with hepatitis C virus consists of combination therapy with (pegylated) interferon-alpha and ribavirin. 1,5-Anhydrohexitol nucleoside analogues are constrained congeners known to mimic the ribonucleoside conformation. Within this series some analogues are
Jong Hyun Cho et al.
Journal of medicinal chemistry, 49(3), 1140-1148 (2006-02-03)
A practical and convenient methodology for the synthesis of chiral cyclopentenol derivative (+)-12a has been developed as the key intermediate that was utilized for the synthesis of biologically active carbocyclic nucleosides. The selective protection of allylic hydroxyl group followed by
Methyl 1H-1,2,4-triazole-3-carboxylate.
Guo XH and Wang QX.
Acta Crystallographica Section E, Structure Reports Online, 61(10), o3217-o3218 (2005)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service