245615
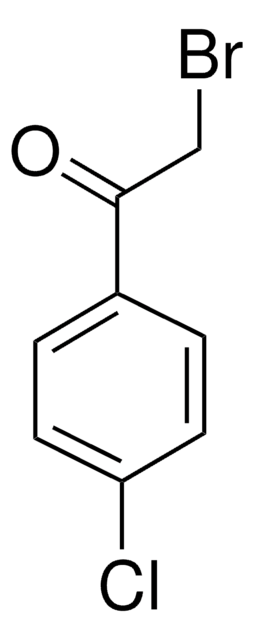
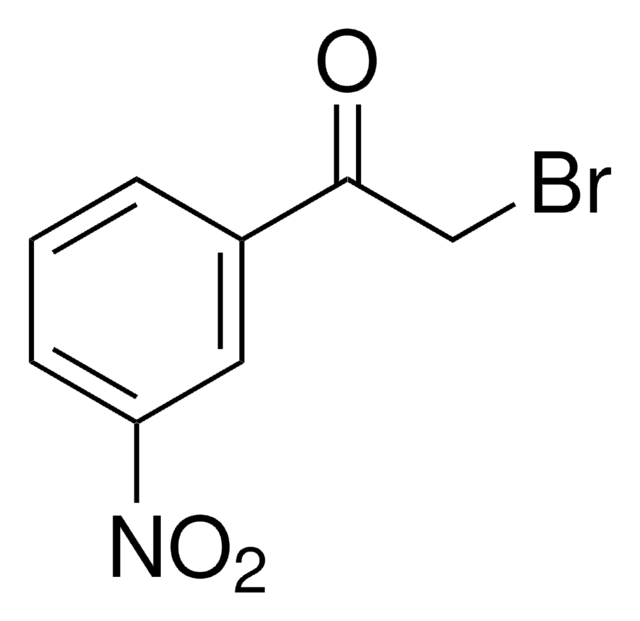
2-Bromo-4′-nitroacetophenone
95%
Synonym(s):
ω-Bromo-4-nitroacetophenone, 4-Nitrophenacyl bromide
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
O2NC6H4COCH2Br
CAS Number:
Molecular Weight:
244.04
Beilstein:
393567
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
95%
mp
94-99 °C (lit.)
functional group
bromo
SMILES string
[O-][N+](=O)c1ccc(cc1)C(=O)CBr
InChI
1S/C8H6BrNO3/c9-5-8(11)6-1-3-7(4-2-6)10(12)13/h1-4H,5H2
InChI key
MBUPVGIGAMCMBT-UHFFFAOYSA-N
Gene Information
human ... PTPN6(5777)
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
2-Bromo-4′-nitroacetophenone was used to study the pKa of the histidine-34 imidazole.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
R R Annand et al.
Biochemistry, 35(14), 4591-4601 (1996-04-09)
In bee venom phospholipase A2, histidine-34 probably functions as a Brønsted base to deprotonate the attacking water. Aspartate-64 and tyrosine-87 form a hydrogen bonding network with histidine-34. We have prepared mutants at these positions and studied their kinetic properties. The
Mechanism of inactivation of rat liver microsomal cytochrome P-450c by 2-bromo-4'-nitroacetophenone.
A Parkinson et al.
The Journal of biological chemistry, 261(25), 11487-11495 (1986-09-05)
The mechanism by which 2-bromo-4'-nitroacetophenone (BrNAP) inactivates cytochrome P-450c, which involves alkylation primarily at Cys-292, is shown in the present study to involve an uncoupling of NADPH utilization and oxygen consumption from product formation. Alkylation of cytochrome P-450c with BrNAP
Koji Karasawa et al.
Analytica chimica acta, 954, 151-158 (2017-01-14)
Five components (hydrogen peroxide, methylglyoxal, dihydroxyacetone, fructose and glucose) of New Zealand manuka honey (Leptospermum scoparium) were analyzed using lucigenin chemiluminescence high-performance liquid chromatography (lucigenin-CL-HPLC). We focused on active oxygen species produced from the components in order to easily detect
M Ungureanu et al.
Revista medico-chirurgicala a Societatii de Medici si Naturalisti din Iasi, 94(1), 157-160 (1990-01-01)
Continuing to present the results of the investigations carried out on the amphoteric ions intermediates of the N-heteroatomic system with 2 natrium atoms in positions 1, 4, new pyrasine derivatives synthetized with p-nitro-phenacyl bromide are described. The observations on their
A Parkinson et al.
The Journal of biological chemistry, 261(25), 11478-11486 (1986-09-05)
The alkylating agent 2-bromo-4'-nitroacetophenone (BrNAP) binds covalently to each of 10 isozymes of purified rat liver microsomal cytochrome P-450 (P-450a-P-450j) but substantially inhibits the catalytic activity of only cytochrome P-450c. Regardless of pH, incubation time, presence of detergents, or concentration
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service