P5057
Paromomycin sulfate salt
powder, suitable for cell culture, BioReagent
About This Item
Productos recomendados
Nombre del producto
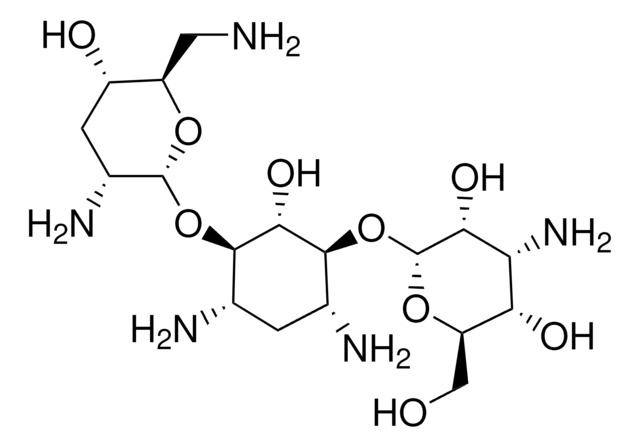
Paromomycin sulfate salt, powder, BioReagent, suitable for cell culture, potency: ≥675 μg per mg
product line
BioReagent
Quality Level
form
powder
potency
≥675 μg per mg
technique(s)
cell culture | mammalian: suitable
antibiotic activity spectrum
Gram-negative bacteria
Gram-positive bacteria
mode of action
protein synthesis | interferes
SMILES string
O[C@H]1[C@H](O)[C@@H](CO)O[C@H](O[C@@]2([H])[C@H](O[C@@]3([H])[C@H](O)[C@H](O[C@]4([H])[C@H](N)[C@@H](O)[C@H](O)[C@H](CN)O4)[C@@H](CO)O3)[C@@H](O)[C@H](N)C[C@@H]2N)[C@@H]1N.C
InChI
1S/C23H45N5O14.CH4/c24-2-7-13(32)15(34)10(27)21(37-7)41-19-9(4-30)39-23(17(19)36)42-20-12(31)5(25)1-6(26)18(20)40-22-11(28)16(35)14(33)8(3-29)38-22;/h5-23,29-36H,1-4,24-28H2;1H4/t5-,6+,7+,8-,9-,10-,11-,12+,13-,14-,15-,16-,17-,18-,19-,20-,21-,22-,23+;/m1./s1
InChI key
OYJABWUHUYVDMJ-UDXJMMFXSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
Application
- Paromomycin sulfate salt has been used as a:
- reference compound in antileishmanial activity
- RNA-binding ligand and interacts with aptamer. This interaction prevents the binding (and cutting) of dicer to RNA duplex.
Biochem/physiol Actions
Mode of Action: Inhibits initiation and elongation during protein synthesis.
Other Notes
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Antibiotic kill curve is a dose response experiment in which mammalian cells are subjected to increasing amounts of selection antibiotic
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico