K1136
Ketorolac tris salt
≥99%, crystalline
Sinónimos:
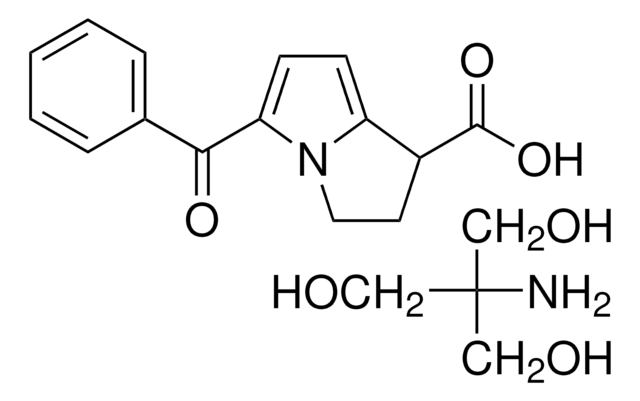
(±)-5-Benzoyl-2,3-dihydro-1H-pyrrolizine-1-carboxylic acid tris salt, Toradol
About This Item
Productos recomendados
biological source
synthetic (organic)
Quality Level
assay
≥99%
form
crystalline
solubility
H2O: soluble 15 mg/mL, clear, colorless to faintly yellow (stable at least one month at −20 °C.)
storage temp.
room temp
SMILES string
NC(CO)(CO)CO.OC(=O)C1CCn2c1ccc2C(=O)c3ccccc3
InChI
1S/C15H13NO3.C4H11NO3/c17-14(10-4-2-1-3-5-10)13-7-6-12-11(15(18)19)8-9-16(12)13;5-4(1-6,2-7)3-8/h1-7,11H,8-9H2,(H,18,19);6-8H,1-3,5H2
InChI key
BWHLPLXXIDYSNW-UHFFFAOYSA-N
Gene Information
human ... PTGS1(5742) , PTGS2(5743)
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
- as an intraperitoneal injection in mice to study the effect of ketorolac on expression of c-Fos (a human proto-oncogene) in ARC (arcuate nucleus of the hypothalamus) POMC (proopiomelanocortin) -EGFP (enhanced green fluorescent protein) neurons
- to treat mice in order to show that this treatment does not prevent IL-1β-mediated inhibition of Agouti-related protein (AgRP) secretion from murine hypothalamic explants
- as an analgesic medication to treat rats induced with acute inflammatory joint injury by injecting carrageenan into the ankle
Biochem/physiol Actions
Caution
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico