D4443
Anti-Derlin-1 antibody produced in rabbit
~1 mg/mL, affinity isolated antibody, buffered aqueous solution
Sinónimos:
Anti-DERtrin 1, Anti-Degradation in endoplasmic reticulum protein 1, Anti-Derl-like protein 1
About This Item
Productos recomendados
biological source
rabbit
conjugate
unconjugated
antibody form
affinity isolated antibody
antibody product type
primary antibodies
clone
polyclonal
form
buffered aqueous solution
mol wt
antigen ~22 kDa
species reactivity
rat, hamster, monkey, mouse, bovine, human, canine
concentration
~1 mg/mL
technique(s)
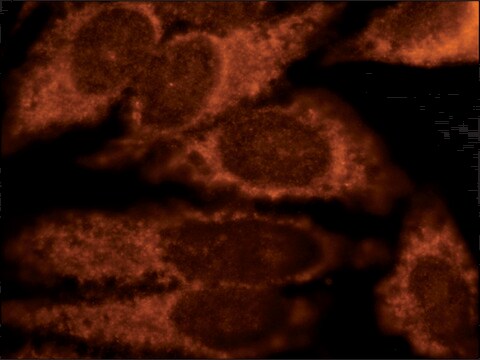
indirect immunofluorescence: 2.5-5 μg/mL using rat NRK cells
western blot (chemiluminescent): 0.2-0.4 μg/mL using whole extract of human HeLa and mouse 3T3 cells.
UniProt accession no.
shipped in
dry ice
storage temp.
−20°C
target post-translational modification
unmodified
Gene Information
human ... DERL1(79139)
mouse ... Derl1(67819)
General description
Immunogen
Application
- immunostaining
- co-immunoprecipitation
- immunofluorescence
Biochem/physiol Actions
Physical form
Disclaimer
¿No encuentra el producto adecuado?
Pruebe nuestro Herramienta de selección de productos.
related product
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico