C9525
4-Chlorouracil
≥99%
Sinónimos:
4-Chloro-2,6-dihydroxypyrimidine, 6-Chloro-2,4-dihydroxypyrimidine, 6-Chlorouracil
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C4H3ClN2O2
Número de CAS:
Peso molecular:
146.53
EC Number:
MDL number:
UNSPSC Code:
41106305
PubChem Substance ID:
NACRES:
NA.51
Productos recomendados
Quality Level
assay
≥99%
form
solid
SMILES string
[H]N1C(Cl)=CC(=O)NC1=O
InChI
1S/C4H3ClN2O2/c5-2-1-3(8)7-4(9)6-2/h1H,(H2,6,7,8,9)
InChI key
PKUFNWPSFCOSLU-UHFFFAOYSA-N
Application
Chlorouracil (4-Chlorouracil; 6-Chlorouracil) is a halogenated uracil that is useful in studies of the effects of halogenation on nucleic acid base-pair stability and alkali metal ion affinity.
Reaction of 6-chlorouracil with 4-(dimethylamino)pyridine, 4-methylpyridine, and pyridin-4-yl-morpholine yielded pyridinium-substituted uracils as chlorides which were converted into pyridinium uracilates by deprotonation. These heterocyclic mesomeric betaines are cross-conjugated and thus possess separate cationic (pyridinium) and anionic (uracilate) moieties. Calculations and X-ray single crystal analyses may be used to characterize these systems and to compare the salts with the betaines.
Reaction of 6-chlorouracil with 4-(dimethylamino)pyridine, 4-methylpyridine, and pyridin-4-yl-morpholine yielded pyridinium-substituted uracils as chlorides which were converted into pyridinium uracilates by deprotonation. These heterocyclic mesomeric betaines are cross-conjugated and thus possess separate cationic (pyridinium) and anionic (uracilate) moieties. Calculations and X-ray single crystal analyses may be used to characterize these systems and to compare the salts with the betaines.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Andreas Schmidt et al.
Organic & biomolecular chemistry, 4(16), 3056-3066 (2006-08-04)
Reaction of 6-chlorouracil with 4-(dimethylamino)pyridine, 4-methylpyridine, and pyridin-4-yl-morpholine yielded pyridinium-substituted uracils as chlorides which were converted into pyridinium uracilates by deprotonation. These heterocyclic mesomeric betaines are cross-conjugated and thus possess separate cationic (pyridinium) and anionic (uracilate) moieties. Calculations and X-ray
Zhibo Yang et al.
Journal of the American Chemical Society, 126(49), 16217-16226 (2004-12-09)
The influence of halogenation on the properties of uracil and its noncovalent interactions with alkali metal ions is investigated both experimentally and theoretically. Bond dissociation energies of alkali metal ion-halouracil complexes, M+(XU), are determined using threshold collision-induced dissociation techniques in
Photochemical transformation of 6-chlorouracil and some alkylated analogues.
Z Kazimierczuk et al.
Biochimica et biophysica acta, 254(2), 157-166 (1971-12-16)
Francesca Bartoccini et al.
Organic & biomolecular chemistry, 10(44), 8860-8867 (2012-10-11)
A small library of 8-substituted 9-deazaxanthines has been prepared by late-stage diversification of an 8-bromo-9-deazaxanthine. By utilizing palladium-catalyzed cross-coupling reactions a single key precursor can be transformed into a variety of 8-substituted-9-deazaxanthine compounds. Three key 8-bromo-9-deazaxanthine intermediates were efficiently prepared
Tumor uptake of radiolabelled pyrimidine bases and pyrimidine nucleosides in animal models--V. 6-[36Cl]chlorouracil, 6-[82Br]bromouracil and 6-[123I]iodouracil.
Y W Lee et al.
International journal of nuclear medicine and biology, 11(3-4), 262-266 (1984-01-01)
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico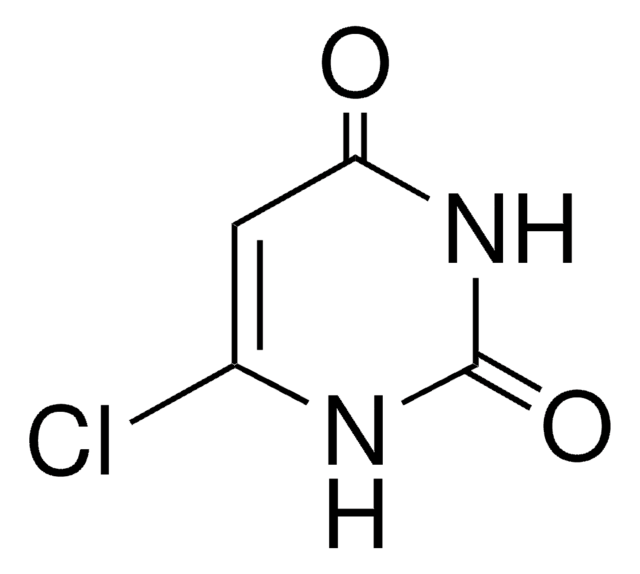
![6-Chloroimidazo[1,2-b]pyridazine hydrochloride AldrichCPR](/deepweb/assets/sigmaaldrich/product/structures/318/484/01bb1292-cad3-4f2f-86cf-0721eb3116d7/640/01bb1292-cad3-4f2f-86cf-0721eb3116d7.png)