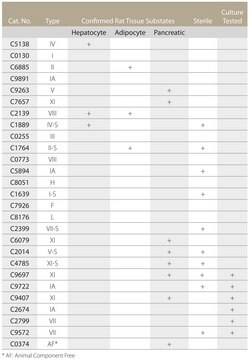
C2799
Colagenasa from Clostridium histolyticum
powder, suitable for cell culture, ≥4 FALGPA units/mg solid, high purity, ≥700 CDU/mg solid (CDU = collagen digestion units)
Sinónimos:
Clostridiopeptidasa A
About This Item
Productos recomendados
biological source
Clostridium histolyticum
Quality Level
form
powder
specific activity
≥4 FALGPA units/mg solid
≥700 CDU/mg solid (CDU = collagen digestion units)
mol wt
68-130 kDa
purified by
chromatography
technique(s)
cell culture | mammalian: suitable
pH
7.4
foreign activity
neutral protease and clostripain ≤1 unit/mg solid
shipped in
dry ice
storage temp.
−20°C
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
Biochem/physiol Actions
Caution
Unit Definition
Preparation Note
Related product
substrate
signalword
Danger
hcodes
Hazard Classifications
Eye Irrit. 2 - Resp. Sens. 1 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico