8.55046
IIDQ-polystyrene
Novabiochem®
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
UNSPSC Code:
13111023
Productos recomendados
Quality Level
product line
Novabiochem®
form
beads
reaction suitability
reaction type: solution phase peptide synthesis
manufacturer/tradename
Novabiochem®
application(s)
peptide synthesis
storage temp.
2-8°C
Categorías relacionadas
General description
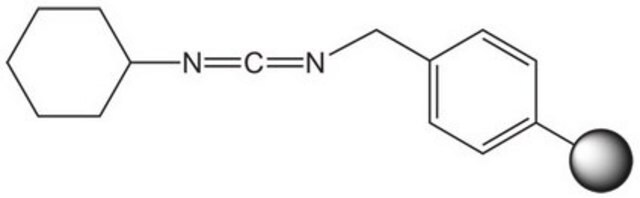
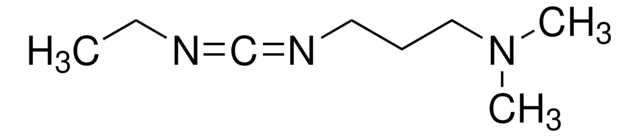
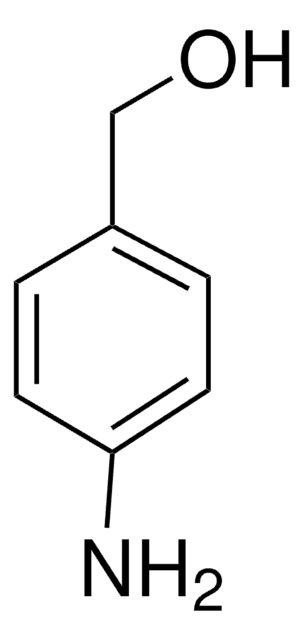
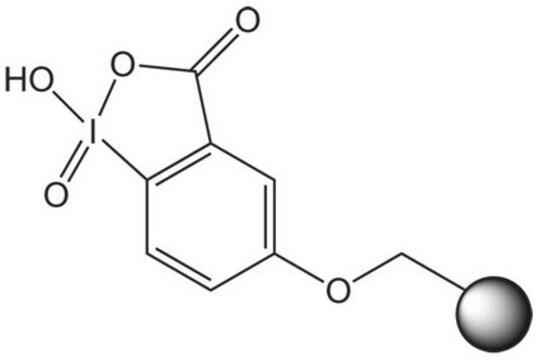
IIDQ-polystyrene (IIDQ-PS) [1] is a polymer-supported version of the IIDQ coupling reagent [2]. IIDQ has many advantages over conventional carbodiimide- or uronium-based reagents: no preactivation step is required, and acid, amine and coupling reagent can be added in any order; in contrast to uronium-based reagents like HBTU, it cannot form guanidinium by-products; and it is totally stable to base. The treatment of a carboxylic acid with IIDQ-PS in DCM or MeCN rapidly generates in situ the corresponding isobutoxycarbonyl mixed anhydride [3]. Attack by nucleophiles preferentially takes place at the less hindered and more electrophilic carbonyl of the carboxylic acid moiety, releasing only volatile carbon dioxide and isobutanol as by-products. If reaction is carried out in the presence of an amine, amide bond formation occurs concurrently with generation of the anhydride. Alternatively, addition of NaBH4 or polymer-supported borohydride to the anhydride will lead directly to the corresponding alcohol. IIDQ-PS appears to be particularly effective for mediating the acylation for anilines, and has also been found to couple peptide fragments without epimerization. In a comparative study, IIDQ-PS was found to give higher yields and greater purities than HATU, EDC-PS or DCC-PS [4]. Occasionally, with some secondary amines the formation of isobutyl carbamate by-products has been observed, resulting from attack by the amine at the carbonyl group.
Literature references
[1] E. Valeur, et al. (2005) Chem. Commun., 1164.
[2] Y. Kiso, et al. (1973) Chem. Pharm. Bull., 21, 2507.
[3] J. R. Vaughan (1951) J. Am. Chem. Soc., 73, 3547.
[4] E. Valeur & M. Bradley, unpublished results.
Literature references
[1] E. Valeur, et al. (2005) Chem. Commun., 1164.
[2] Y. Kiso, et al. (1973) Chem. Pharm. Bull., 21, 2507.
[3] J. R. Vaughan (1951) J. Am. Chem. Soc., 73, 3547.
[4] E. Valeur & M. Bradley, unpublished results.
Linkage
Replaces: 01-64-0469
Analysis Note
Color (visual): yellow to beige to amber
Appearance of substance (visual): beads
Loading (determined by HPLC after reaction of 3-phenylpropionic acid with cyclohexlamine): 1.3 - 1.9 mmol/g
Swelling Volume (in DMF): lot specific result
The polymer matrix is copoly (styrene-1 % DVB) 200 - 400 mesh.
Appearance of substance (visual): beads
Loading (determined by HPLC after reaction of 3-phenylpropionic acid with cyclohexlamine): 1.3 - 1.9 mmol/g
Swelling Volume (in DMF): lot specific result
The polymer matrix is copoly (styrene-1 % DVB) 200 - 400 mesh.
Legal Information
Novabiochem is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico