O3750
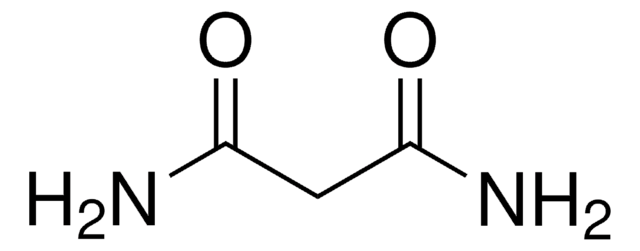
Oxamic acid
≥98%
Sinónimos:
Aminooxoacetic acid, Oxalic acid monoamide
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
NH2COCO2H
Número de CAS:
Peso molecular:
89.05
Beilstein/REAXYS Number:
1743294
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
assay
≥98%
form
powder
mp
207-210 °C (dec.) (lit.)
SMILES string
NC(=O)C(O)=O
InChI
1S/C2H3NO3/c3-1(4)2(5)6/h(H2,3,4)(H,5,6)
InChI key
SOWBFZRMHSNYGE-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
Oxamic acid (OA) can be used as a reactant to prepare 6-phenanthridinecarboxamide by direct C-H carbamoylation reaction using ammonium persulfate in DMSO. It can also be used as an organic ligand to prepare functionalized metal oxide nanoparticles for various biological applications. OA along with p-aminobenzoic acid is used to functionalize Au nanoparticles for the development of a sensor to detect Fe3+ ions by the calorimetric method.
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Chrysi Xintaropoulou et al.
BMC cancer, 18(1), 636-636 (2018-06-06)
Novel therapeutic approaches are required to treat ovarian cancer and dependency on glycolysis may provide new targets for treatment. This study sought to investigate the variation of expression of molecular components (GLUT1, HKII, PKM2, LDHA) of the glycolytic pathway in
Adam D Moorhouse et al.
Chemical communications (Cambridge, England), 47(1), 230-232 (2010-08-03)
hLDH-5 has emerged as a promising target for anti-glycolytic cancer chemotherapy. Here we report a first generation of bifunctional inhibitors, which show promising activity against hLDH-5.
Richard A Ward et al.
Journal of medicinal chemistry, 55(7), 3285-3306 (2012-03-16)
Lactate dehydrogenase A (LDHA) catalyzes the conversion of pyruvate to lactate, utilizing NADH as a cofactor. It has been identified as a potential therapeutic target in the area of cancer metabolism. In this manuscript we report our progress using fragment-based
Metal-, Photocatalyst-, and Light-Free Direct C-H Acylation and Carbamoylation of Heterocycles
Westwood MT, et al.
Organic Letters, 21(17), 7119-7123 (2019)
Xuguang Yang et al.
Cancer immunology research, 8(11), 1440-1451 (2020-09-13)
The mechanisms responsible for radioresistance in pancreatic cancer have yet to be elucidated, and the suppressive tumor immune microenvironment must be considered. We investigated whether the radiotherapy-augmented Warburg effect helped myeloid cells acquire an immunosuppressive phenotype, resulting in limited treatment
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico