476994
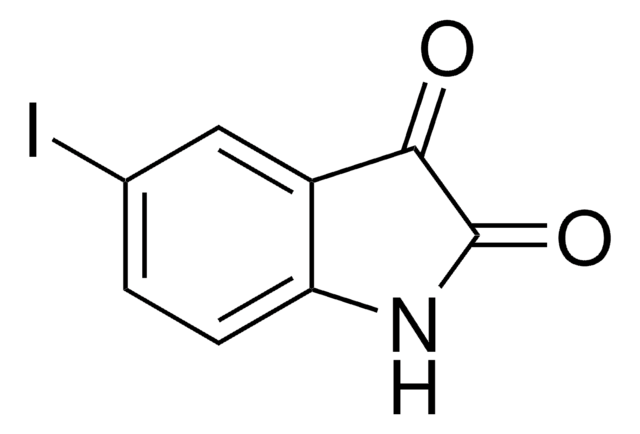
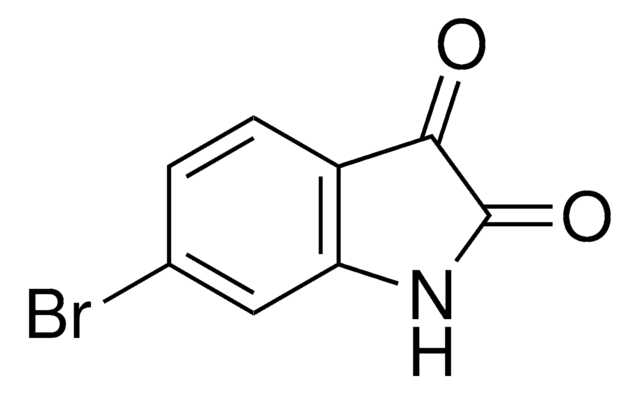
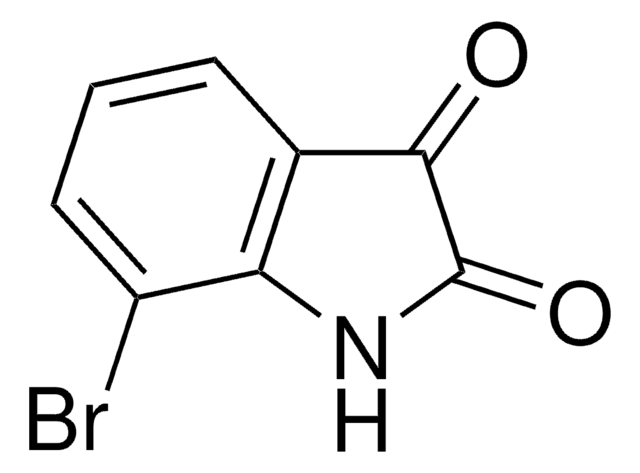
5-Bromoisatin
technical grade, 90%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C8H4BrNO2
Número de CAS:
Peso molecular:
226.03
Beilstein/REAXYS Number:
383760
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
grade
technical grade
Quality Level
assay
90%
impurities
<10% isatin
mp
247-252 °C (lit.)
functional group
bromo
ketone
SMILES string
Brc1ccc2NC(=O)C(=O)c2c1
InChI
1S/C8H4BrNO2/c9-4-1-2-6-5(3-4)7(11)8(12)10-6/h1-3H,(H,10,11,12)
InChI key
MBVCESWADCIXJN-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
5-Bromoisatin is a 5-haloisatin. One of the methods reported for its synthesis is by reacting N-halosaccharins with isatin in the presence of SiO2. Its inotropic activity has been studied on rhythmically stimulated papillary muscles of guinea pigs. It is reported to exhibit analgesic and sedative properties at a dose of 0.2g/kg in mice.
Application
5-Bromoisatin may be used in the synthesis of the following:
- N-derivatives of 5-bromoisatin
- N-substituted pyrroles
- linear polyaryleneoxindoles
- 5-bromodioxindole
- cinchoninic acid derivatives
- 3-hydroxyoxindole
- S-benzyldithiocarbazate Schiff Bases
- 5-bromooxindole
- Morita-Baylis-Hillman adducts of isatin derivatives
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Possible psychopharmacological agents. X. Synthesis of Some Fluorine Containing Indole-2, 3-dione Derivatives.
Joshi KC, et al.
J. Prakt. Chem., 322(2), 314-320 (1980)
V N Garalene et al.
Voprosy meditsinskoi khimii, 30(5), 56-59 (1984-09-01)
Effect of isatin and of three its derivatives on content of lactate, pyruvate and glycogen was studied in rat tissues. The substances studied, except of 5-butylisatin, decreased the content of lactic acid with simultaneous increase of glycogen content in liver
A facile one-pot method for the preparation of N-alkyl isatins under microwave irradiation.
Azizian J, et al.
Synthetic Communications, 33(5), 789-793 (2003)
Synthesis, characterization and cytotoxic activity of S-benzyldithiocarbazate Schiff bases derived from 5-fluoroisatin, 5-chloroisatin, 5-bromoisatin and their crystal structures.
Manan MAFA, et al.
Journal of Chemical Crystallography, 41(11), 1630-1641 (2011)
A practical large-scale preparation of 5'-bromospiro (cyclohexane-1,3-[3H] indol)-2'(1'H)-one.
Wilk BK, et al.
Organic preparations and procedures international, 37(3), 283-285 (2005)
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico