389412
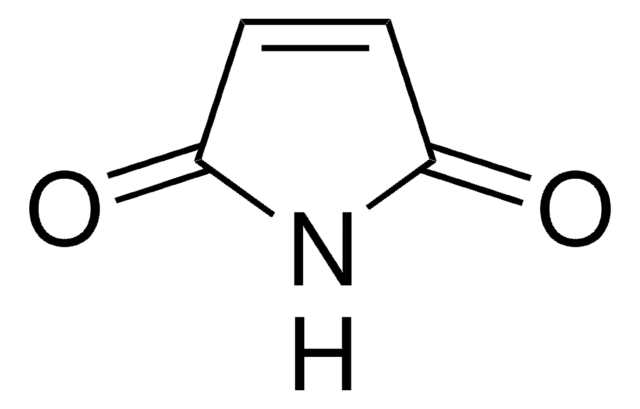
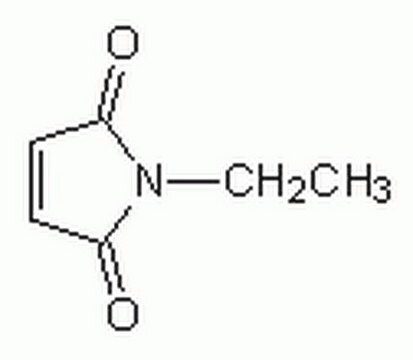
N-Methylmaleimide
97%
Sinónimos:
1-Methyl-1H-pyrrole-2,5-dione, 1-Methyl-2,5-dihydro-1H-pyrrole-2,5-dione, 1-Methylpyrrole-2,5-dione, N-Methyl maleic imide, N-Methylpyrrole-2,5-dione
About This Item
Productos recomendados
Quality Level
assay
97%
form
powder
mp
94-96 °C (lit.)
SMILES string
CN1C(=O)C=CC1=O
InChI
1S/C5H5NO2/c1-6-4(7)2-3-5(6)8/h2-3H,1H3
InChI key
SEEYREPSKCQBBF-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
- In the synthesis of organic structure directing agents for designing silicogermanate zeolites.
- As a dienophile in Diels-Alder reaction.
- For the synthesis of biologically active pyrrolo[2,1-a]isoquinolines.
- As a dipolarophile in [3 + 2] dipolar addition reactions.
- As a co-monomer in atom transfer radical polymerization of styrene to obtain styrene copolymers with programmed microstructure.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Skin Corr. 1B - Skin Sens. 1
Storage Class
8A - Combustible corrosive hazardous materials
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico