330752
Triphosgene
reagent grade, 98%
Sinónimos:
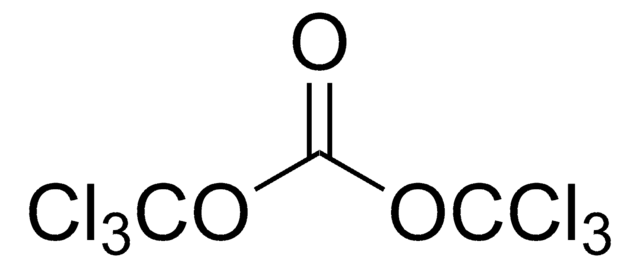
Bis(trichloromethyl) carbonate
About This Item
Productos recomendados
grade
reagent grade
Quality Level
assay
98%
form
powder
reaction suitability
reaction type: Coupling Reactions
bp
203-206 °C (lit.)
mp
79-83 °C (lit.)
functional group
carbonate
chloro
storage temp.
2-8°C
SMILES string
ClC(Cl)(Cl)OC(=O)OC(Cl)(Cl)Cl
InChI
1S/C3Cl6O3/c4-2(5,6)11-1(10)12-3(7,8)9
InChI key
UCPYLLCMEDAXFR-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
It is also commonly employed as a coupling agent in the synthesis of carbonyl compounds.
Application
- Thiocarbonates from thiols and alcohols by one-pot, three-component reaction.
- Substituted azetidin-2-ones from acids and imines via ketene–imine cycloaddition reaction.
- Methyl (S)-2-isocyanato-3-phenylpropanoate from L-phenylalanine methyl ester hydrochloride in the presence of sodium bicarbonate.
- Acyl azides derivatives from various carboxylic acids and sodium azide.
- Immunosuppressant agent cyclosporin by solid-phase peptide synthesis.
- Allyl azides from allyl alcohols and sodium azide in one pot method.
- Esterification coupling reagent di-2-thienyl carbonate, from 2(5H)-thiophenone.
- 2-Chloronicotinaldehydes via cyclization of the corresponding enamides.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 1 Inhalation - Skin Corr. 1B
Storage Class
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Faceshields, Gloves, type P3 (EN 143) respirator cartridges
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico