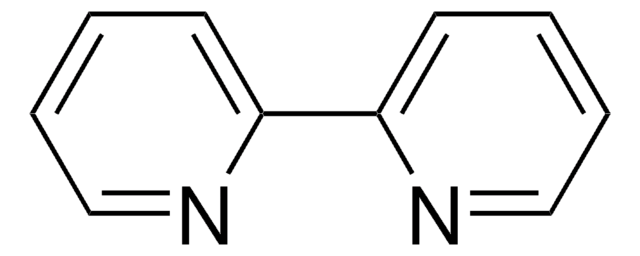
301868
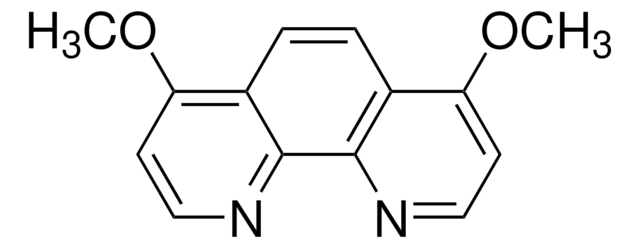
4,7-Phenanthroline
98%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C12H8N2
Número de CAS:
Peso molecular:
180.21
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
assay
98%
mp
172-174 °C (lit.)
SMILES string
c1cnc2ccc3ncccc3c2c1
InChI
1S/C12H8N2/c1-3-9-10-4-2-8-14-12(10)6-5-11(9)13-7-1/h1-8H
InChI key
DATYUTWESAKQQM-UHFFFAOYSA-N
Categorías relacionadas
General description
4,7-Phenanthroline reacts with ruthenium carbonyl to yield cyclometalated derivatives.
Application
4,7-Phenanthroline was used in preparation of:
- cyclic tetranuclear half-sandwich ruthenium(II) complexes
- positively charged homochiral cyclic trinuclear metallacalix[3]arene species
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Julien Frey et al.
Journal of the American Chemical Society, 130(33), 11013-11022 (2008-07-26)
Variously substituted coordinating rigid rods have been synthesized which incorporate a central 4,7-phenanthroline nucleus attached to two 2-pyridyl groups via its 3 and 8 positions, so as to yield bis-bidentate chelates, the two-coordinating axes of the chelates being parallel to
Miguel A Galindo et al.
Journal of inorganic biochemistry, 102(5-6), 1025-1032 (2008-01-12)
The reaction between [(eta(6)-p-cymene)Ru(H2O)3]X2 and 4,7-phenanthroline (phen) leads to the formation of the rectangular tetranuclear complexes [(eta(6)-p-cymene)4Ru4(mu-4,7-phen-N4,N7)(2)(mu-OH)4]X4 (X=NO3, 1a; SO3CF3, 1b) which have been structurally characterised by X-ray crystallography. 1H NMR spectroscopic studies suggest the presence of a partially dissociated
Miguel A Galindo et al.
Dalton transactions (Cambridge, England : 2003), (10)(10), 1563-1566 (2004-07-15)
Reaction of [(dach)Pd(NO3)2] entities (dach = (R,R)-1,2-diaminocyclohexane, (S,S)-1,2-diaminocyclohexane) and 4,7-phenanthroline (phen) providing, respectively, 90 and 120 degrees bond angles, leads to the formation of two novel positively charged homochiral cyclic trinuclear metallacalix[3]arene species [((R,R)-1,2-diaminocyclohexane)Pd(phen)]3(NO3)6 (2a) and [((S,S)-1,2-diaminocyclohexane)Pd(phen)]3(NO3)6 (2b). These species
L W Mitchell et al.
Archives of biochemistry and biophysics, 300(1), 169-177 (1993-01-01)
Porphobilinogen synthase (PBGS) is essential to all life forms; in mammals it is definitively established that Zn(II) is required for activity. The literature regarding the metal requirement for PBGS in other animals, plants, and bacteria neither establishes nor disproves a
V Arluison et al.
Biochemistry, 37(20), 7268-7276 (1998-06-04)
RNA:pseudouridine synthetase (Pus1) from Saccharomyces cerevisiae is a multisite specific enzyme that catalyzes the formation of pseudouridine at positions 34 and 36 of intron-containing precursor tRNAIle and at positions 27 and/or 28 of several yeast tRNAs. In this paper we
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico

![Pyrazino[2,3-f][1,10]phenanthroline 99% (HPLC)](/deepweb/assets/sigmaaldrich/product/structures/226/341/31d3909e-6700-4a3e-bfb3-9ed1f6b66ee2/640/31d3909e-6700-4a3e-bfb3-9ed1f6b66ee2.png)