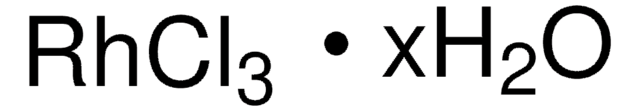199982
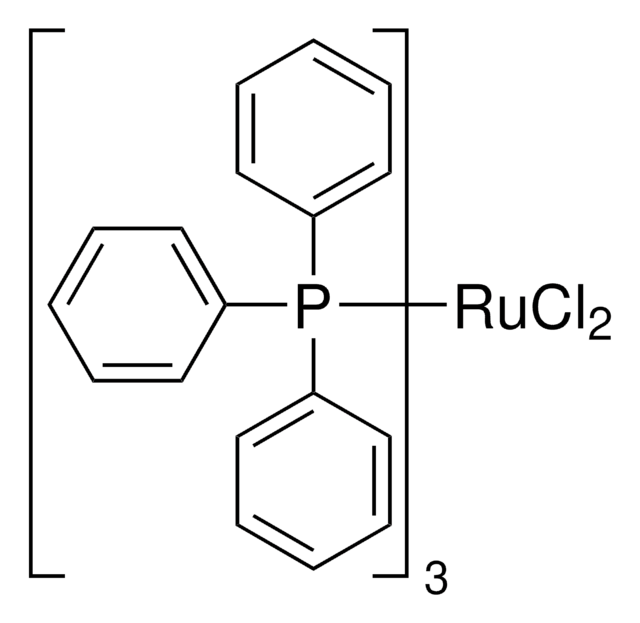
Tris(triphenylphosphine)rhodium(I) chloride
Sinónimos:
NSC 124140, RhCl(PPh3)3, Rhodium(I) tris(triphenylphosphine) chloride, Wilkinson’s catalyst
About This Item
Productos recomendados
reaction suitability
core: rhodium
reagent type: catalyst
SMILES string
Cl[Rh].c1ccc(cc1)P(c2ccccc2)c3ccccc3.c4ccc(cc4)P(c5ccccc5)c6ccccc6.c7ccc(cc7)P(c8ccccc8)c9ccccc9
InChI
1S/3C18H15P.ClH.Rh/c3*1-4-10-16(11-5-1)19(17-12-6-2-7-13-17)18-14-8-3-9-15-18;;/h3*1-15H;1H;/q;;;;+1/p-1
InChI key
IXAYKDDZKIZSPV-UHFFFAOYSA-M
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
Catalyst used for many organic reactions including:
- Chemoselective allylic alkylations
- Stoichiometric activation of Si-H bonds and hydrosilylations
- Inter- and intramolecular hydroacylation of alkenes along with a cocatalyst
- Polymerization of diorganostannanes
signalword
Warning
hcodes
Hazard Classifications
Aquatic Chronic 4 - Skin Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Vinyl-metal reagents play a pivotal role in organic synthesis. Among the vinyl-metal reagents available, silicon-based reagents are of increasing importance. This is largely due to their low cost, minimal toxicity, ease of handling, and the simplicity of byproduct removal.
A Highly Efficient Hydrosilylation Catalyst, sigma-aldrich is pleased to offer [Cp*Ru(MeCN)3]PF6, as well as a number of other catalysts for hydrosilylation.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico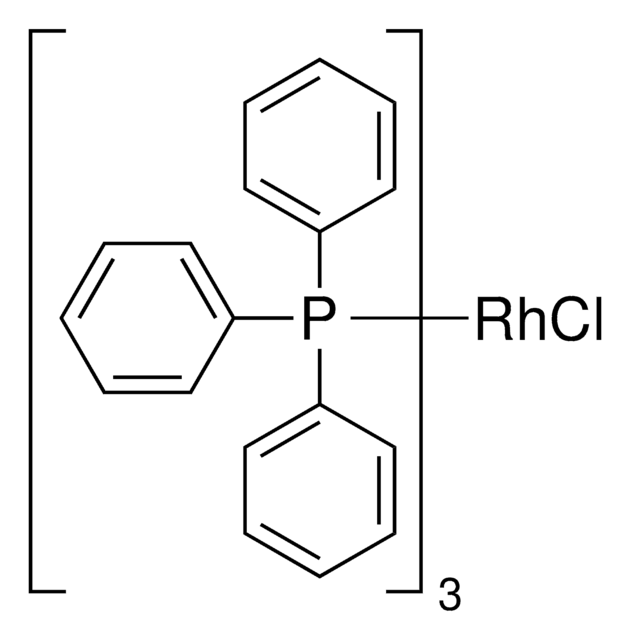

rhodium(I) tetrafluoroborate 98%](/deepweb/assets/sigmaaldrich/product/structures/138/264/047825b4-1f5a-486a-9f51-8d9c18b1382f/640/047825b4-1f5a-486a-9f51-8d9c18b1382f.png)

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)