163392
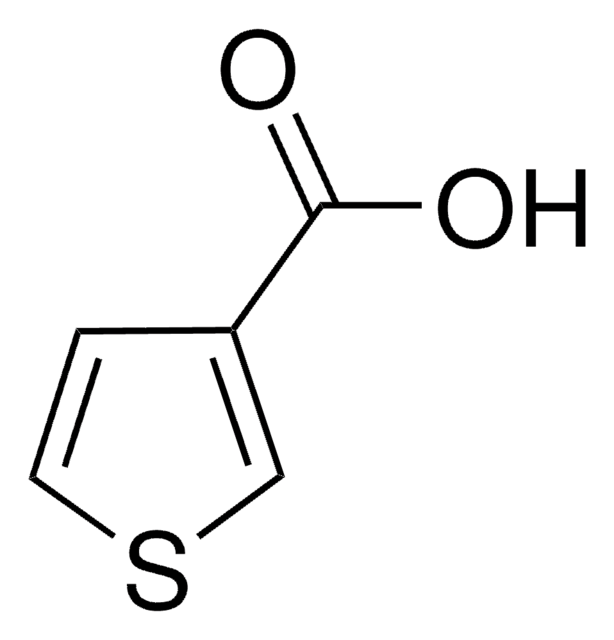
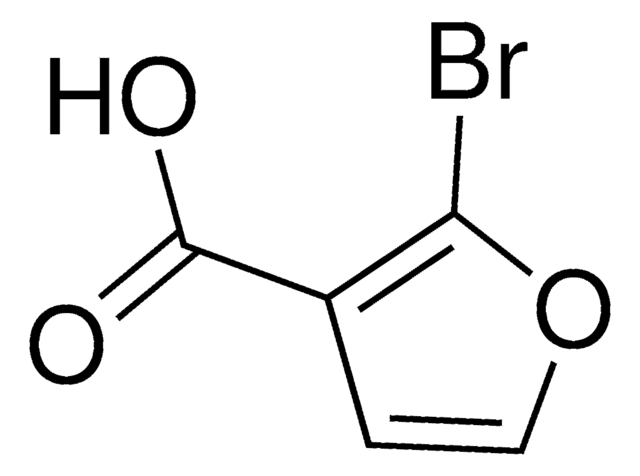
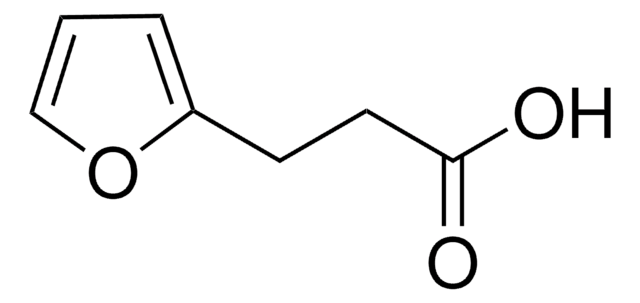
3-Furoic acid
98%
Sinónimos:
Furan-3-carboxylic acid
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C5H4O3
Número de CAS:
Peso molecular:
112.08
Beilstein/REAXYS Number:
108638
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
assay
98%
mp
120-122 °C (lit.)
functional group
carboxylic acid
SMILES string
OC(=O)c1ccoc1
InChI
1S/C5H4O3/c6-5(7)4-1-2-8-3-4/h1-3H,(H,6,7)
InChI key
IHCCAYCGZOLTEU-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Application
3-Furoic acid can be used as a reactant to synthesize:
- Furan-2,5- and furan-2,4-dicarboxylic acid under solvent-free conditions via disproportionation reaction.
- (±)-Hyperolactone A by reacting with 2-methylbutanal.
- Furo[2,3-b]pyridin-4-one-5-carboxylate ester derivatives as potential non-nucleoside inhibitors of human herpesvirus polymerases.
Biochem/physiol Actions
3-Furoic acid exhibits hypolipidemic activity in rodents. It lowers serum cholesterol and serum triglyceride levels in mice and rats.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
I H Hall et al.
Pharmaceutical research, 2(5), 233-238 (1985-09-01)
2-Furoic acid, 3-furoic acid, 3,4-furan dicarboxylic acid and furyl-acrylic acid were evaluated for hypolipidemic activity in mice and rats. 2-Furoic acid was the most potent agent of the four tested, lowering serum cholesterol levels 41 % and serum triglyceride levels
Concurrent formation of furan-2, 5-and furan-2, 4-dicarboxylic acid: unexpected aspects of the Henkel reaction.
Thiyagarajan S, et al.
Royal Society of Chemistry Advances, 3(36), 15678-15686 (2013)
H M Liebich et al.
Journal of chromatography, 525(1), 1-14 (1990-01-26)
Altogether 143 of the organic acids regularly occurring in urine of healthy individuals are identified as methyl esters by gas chromatography-mass spectrometry with respect to their complete chemical structures. They are classified as dicarboxylic acids, oxocarboxylic acids, hydroxycarboxylic acids, aromatic
M J Nozal et al.
Journal of chromatography. A, 917(1-2), 95-103 (2001-06-14)
A high-performance liquid chromatographic method for determining 5-hydroxymethyl-2-furaldehyde (hydroxymethylfurfural), 2-furaldehyde (furfural), furan-2-carboxylic acid (2-furoic acid), furan-3-carboxylic acid (3-furoic acid), furan-3-carboxaldehyde (3-furaldehyde) and 2-aminobenzoic acid methyl ester (methyl anthranilate) in honey and honeydew samples is described. To prevent matrix interference and
Sorin Tunaru et al.
Nature medicine, 9(3), 352-355 (2003-02-04)
Nicotinic acid (niacin), a vitamin of the B complex, has been used for almost 50 years as a lipid-lowering drug. The pharmacological effect of nicotinic acid requires doses that are much higher than those provided by a normal diet. Its
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico