All Photos(2)
About This Item
Empirical Formula (Hill Notation):
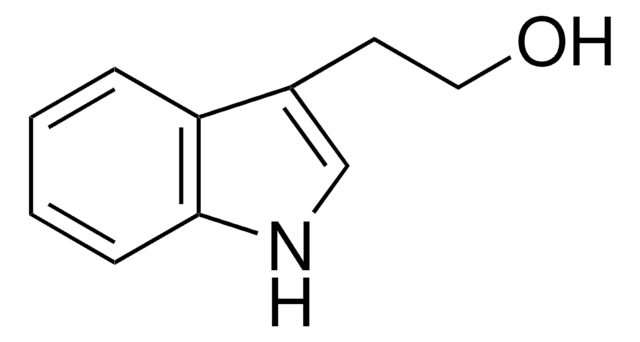
C9H9NO
CAS Number:
Molecular Weight:
147.17
EC Number:
MDL number:
UNSPSC Code:
12352005
PubChem Substance ID:
NACRES:
NA.25
Recommended Products
mp
96-99 °C (lit.)
storage temp.
2-8°C
SMILES string
OCc1c[nH]c2ccccc12
InChI
1S/C9H9NO/c11-6-7-5-10-9-4-2-1-3-8(7)9/h1-5,10-11H,6H2
InChI key
IVYPNXXAYMYVSP-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Indole-3-carbinol is a novel secondary plant metabolite produced in cruciferous vegetables, such as cabbage, cauliflower and brussels sprouts.
Application
Indole-3-carbinol has been used for encapsulation with poly-lactic-co-glycolic acid (PLGA), to study its in-vitro anti-cancerogenic effects on breast adenocarcinoma epithelial (MCF7), colon adenocarcinoma epithelial (Caco2), prostate carcinoma epithelial (PC3) cells. It has also been used as a cytochrome P4501A (CYP1A) inducer.
Biochem/physiol Actions
Indole-3-carbinol (I3C) activates aryl hydrocarbon receptor (AhR) and induces G1 cell cycle arrest and apoptosis. Thus, it acts as a potential anti-cancer agent. In addition, it induces estradiol metabolism by stimulating cytochrome P450 enzymes. Therefore, I3C is considered to be a potent chemotherapeutic for various types of cancer including, breast, prostate, colon cancer, and leukemia.
Inhibits cancinogenesis at the initiation stage. Has be shown to inhibit carcinogenesis in several animal species, but it enhances tumor incidence if administered at a post-initiation stage. Found in cruciferous vegetables.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Aromatic hydrocarbon responsiveness-receptor agonists generated from indole-3-carbinol in vitro and in vivo: comparisons with 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin.
Bjeldanes LF, et al.
Proceedings of the National Academy of Sciences of the USA, 88(21), 9543-9547 (1991)
Jing-Ru Weng et al.
Cancer letters, 262(2), 153-163 (2008-03-04)
During the course of oncogenesis and tumor progression, cancer cells constitutively upregulate signaling pathways relevant to cell proliferation and survival as a strategy to overcome genomic instability and acquire resistance phenotype to chemotherapeutic agents. In light of this clinical and
G S Bailey et al.
Journal of the National Cancer Institute, 78(5), 931-934 (1987-05-01)
Indole-3-carbinol (I3C), a natural constituent of cruciferous vegetables, is an inhibitor in several experimental animal models of carcinogenesis by polynuclear aromatic hydrocarbons or aflatoxin B1 (AFB1) when administered prior to or during carcinogen exposure. For assessment of the postinitiation effects
H L Bradlow et al.
Annals of the New York Academy of Sciences, 889, 204-213 (2000-02-11)
Previous studies from this laboratory have suggested that 2-hydroxyestrone is protective against breast cancer, whereas the other principal metabolite, 16 alpha-hydroxyestrone, and the lesser metabolite quantitatively, 4-hydroxyestrone, are potent carcinogens. Attempts to directly decrease the formation of the 16-hydroxylated metabolite
PLGA encapsulation and radioiodination of indole-3-carbinol: investigation of anticancerogenic effects against MCF7, Caco2 and PC3 cells by in vitro assays
Yildiz G, et al.
J. Radioanal. Nucl. Chem., 311(2), 1043-1052 (2017)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service