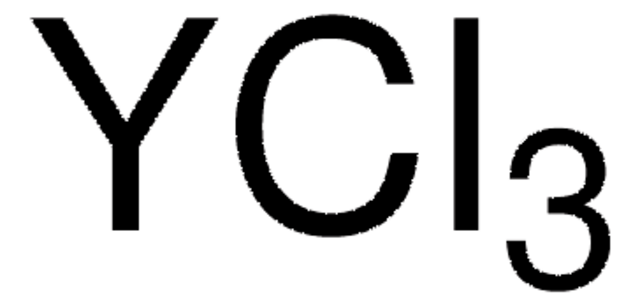429406
Cerium(III) chloride
AnhydroBeads™, −10 mesh, ≥99.99% trace metals basis
Synonym(s):
Cerium trichloride, Cerous chloride
About This Item
synthesis grade
Recommended Products
grade
for synthesis
synthesis grade
Quality Level
product line
AnhydroBeads™
Assay
≥99.99% trace metals basis
form
beads
reaction suitability
reagent type: catalyst
core: cerium
impurities
≤100.0 ppm Trace Rare Earth Analysis
particle size
−10 mesh
mp
848 °C (lit.)
density
3.97 g/mL at 25 °C (lit.)
SMILES string
Cl[Ce](Cl)Cl
InChI
1S/Ce.3ClH/h;3*1H/q+3;;;/p-3
InChI key
VYLVYHXQOHJDJL-UHFFFAOYSA-K
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- The role of dynamic ligand exchange in the oxidation chemistry of cerium (III): This study explores the redox chemistry of cerium, utilizing Cerium(III) chloride in oxidation reactions to understand its potential in various chemical transformations (JR Robinson et al., 2016).
- Photocatalytic C- C bond cleavage and amination of cycloalkanols by cerium (III) chloride complex: Examines the photocatalytic properties of a cerium(III) chloride complex, used for C-C bond cleavage and amination, highlighting its potential in organic synthesis (JJ Guo et al., 2016).
- Dehydration, hydrolysis and oxidation of cerium chloride heptahydrate in air atmosphere: Investigates the behavior of hydrated forms of Cerium(III) chloride under varying conditions, contributing to understanding its stability and reactivity (S Xue et al., 2017).
Other Notes
Legal Information
accessory
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1 - Skin Corr. 1C
Storage Class Code
8B - Non-combustible corrosive hazardous materials
WGK
WGK 2
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
We carry a large variety of electrophiles and nucleophiles that are widely used in C–C bond-forming reactions. This group of products contains many organometallic reagents as well as commonly-used alkylating and acylating reagents.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 429406-25G | 4061838139672 |
| 429406-5G | 4061832105628 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service