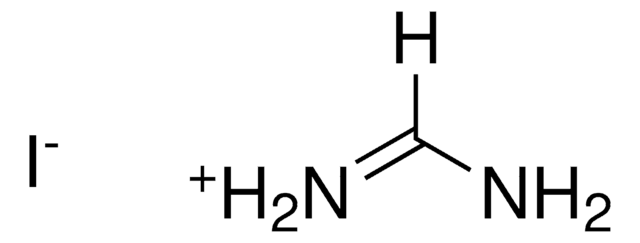
398853
Lead(II) bromide
99.999% trace metals basis
Synonym(s):
Lead dibromide
About This Item
Recommended Products
grade
for analytical purposes
Quality Level
Assay
99.999% trace metals basis
form
powder
reaction suitability
core: lead
impurities
≤15.0 ppm Trace Metal Analysis
bp
892 °C (lit.)
mp
371 °C (lit.)
density
6.66 g/mL at 25 °C (lit.)
SMILES string
Br[PbH2]Br
InChI
1S/2BrH.Pb/h2*1H;/q;;+2/p-2
InChI key
ZASWJUOMEGBQCQ-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
General description
Application
- To prepare an electrolyte for a high performance all-solid-state bromide-ion battery.
- As a precursor to prepare organic–inorganic hybrid perovskite materials for solar cells and light emitting devices (LEDs).
- As a starting material to prepare photocatalytic CsPbBr3@SiO2 composites with good water stability.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Repr. 1A - STOT RE 2
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Since the first report of the low-cost dye-sensitized solar cell (DSSC) in 1991 by Gratzel and his coworker,1 dye-sensitized solar cells (DSSC) has been regarded as one of the most promising photovoltaic technologies because of their transparent and colorful characteristics, as well as low cost.
Colloidal quantum dots (CQDs) are semiconducting crystals of only a few nanometers (ca. 2–12 nm) coated with ligand/surfactant molecules to help prevent agglomeration.
The past several decades have seen major advancements in the synthesis of metal nanomaterials. Most recently, controlled synthesis has become versatile enough to regulate the exact number of atoms and ligands of very small metal nanoparticles, referred to as “clusters”.
Next generation solar cells have the potential to achieve conversion efficiencies beyond the Shockley-Queisser (S-Q) limit while also significantly lowering production costs.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 398853-25G | |
| 398853-5G | 4061831984354 |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service