384216
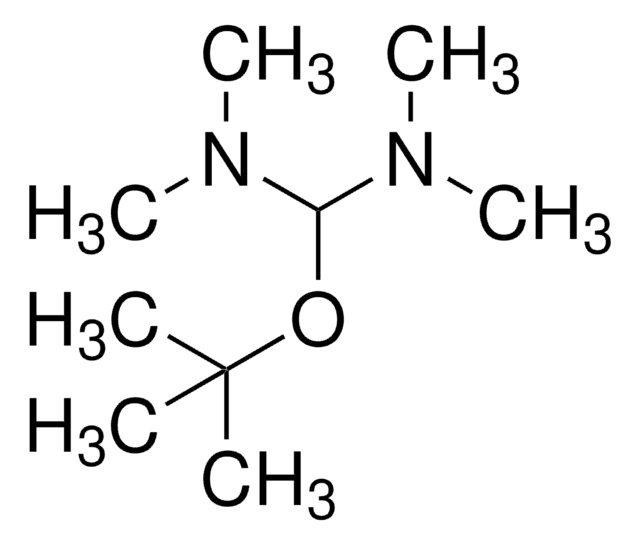
tert-Butoxy bis(dimethylamino)methane
Synonym(s):
Bredereck’s reagent
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(CH3)3COCH[N(CH3)2]2
CAS Number:
Molecular Weight:
174.28
Beilstein:
1901973
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
form
liquid
Quality Level
refractive index
n20/D 1.422 (lit.)
bp
50-55 °C/15 mmHg (lit.)
density
0.844 g/mL at 25 °C (lit.)
functional group
amine
ether
SMILES string
CN(C)C(OC(C)(C)C)N(C)C
InChI
1S/C9H22N2O/c1-9(2,3)12-8(10(4)5)11(6)7/h8H,1-7H3
InChI key
HXRAMSFGUAOAJR-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
tert-Butoxy bis(dimethylamino)methane (Bredereck′s reagent) may be used in the following studies:
- Enamination of active methylene and methyl groups.
- As condensation reagent in the synthesis of benz[c,d]indoles.
- Preparation of methyl and ethyl 3-dimethylamino-2-(indol-3-yl)propenoate.
- Synthesis of the macrolide natural product (-)-gloeosporone.
- Preparation of new bioactive naphthyridine alkaloids lophocladine A and B.
- Preparation of (E/Z)-ethyl 6,7-dichloro-3-[1-cyano-2-(dimethylamino)vinyl]-1-methyl-1H-indole-2-carboxylate.
- Preparation of (E)-ethyl 3-(dimethylamino)-2-(1H-indol-3-yl)acrylate.
- α-Enamination of ketones and esters.
Reactant for:
- Preparation of pyrroloquinazolines as photochemotherapeutic agents
- Enanatioslective formal synthesis of the Cinchona alkaloid quinine via stereoselective intermolecular radical addition
- Synthesis of camptothecin via intramolecular isomuenchnone cycloaddition reaction
- Aminomethylenation reactions
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
105.8 °F - closed cup
Flash Point(C)
41 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Tetrahedron Asymmetry, 17, 1217-1217 (2006)
Benz [c, d] indoles-I. The use of tert-butoxy-bis (dimethylamino) methane as condensation reagent.
Hafliger W and Knecht H.
Tetrahedron Letters, 25(3), 285-288 (1984)
Tetrahedron Letters, 31, 2105-2105 (1990)
Tetrahedron Letters, 48, 613-613 (2007)
Matthias Lotter et al.
Archiv der Pharmazie, 339(12), 677-679 (2006-11-17)
The one-pot reaction of 4-benzylpyridine-3-carbonitrile with Bredereck's reagent and subsequent treatment with either glacial acetic acid and sulfuric acid or ammonium acetate provided the new bioactive naphthyridine alkaloids lophocladine A and B, respectively.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service




![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)