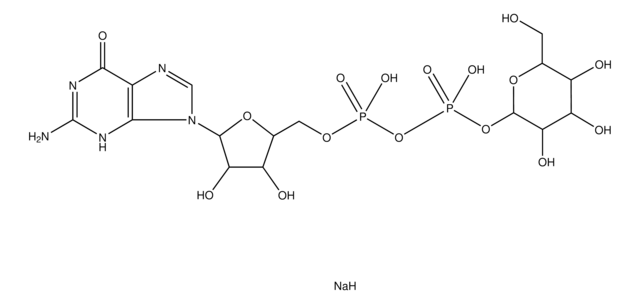
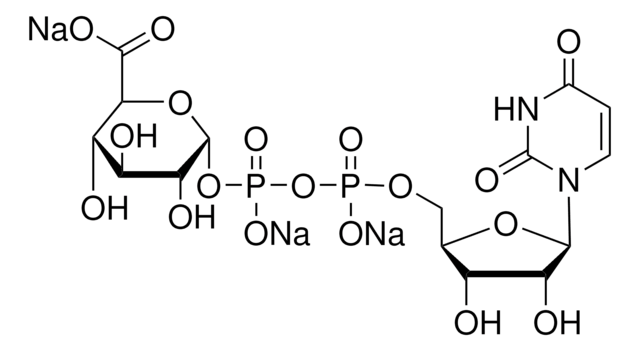
U5252
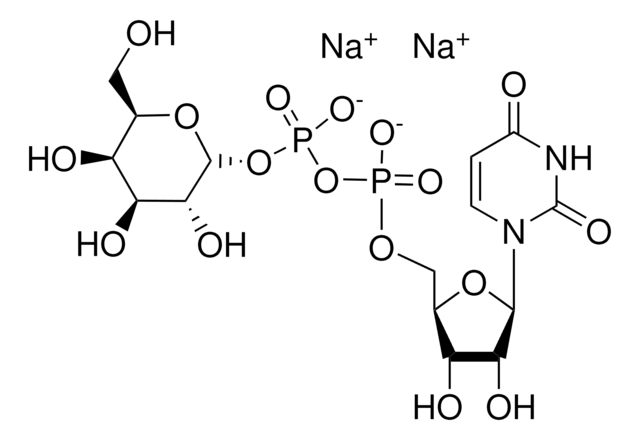
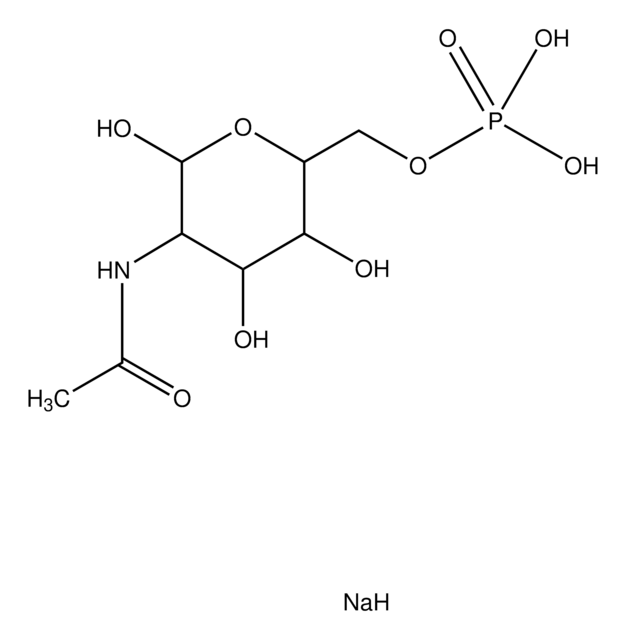
Uridine 5′-diphospho-N-acetylgalactosamine disodium salt
≥97%
Synonym(s):
(UDP)-GalNAc, UDP-GalNAc, UDP-N-acetylgalactosamine, Uridine[5′]diphospho[1](2-acetamino-2-deoxy-α-D-galactopyranose) disodium salt
About This Item
Recommended Products
biological source
synthetic (organic)
Quality Level
Assay
≥97%
form
powder
storage temp.
−20°C
SMILES string
[Na+].[Na+].CC(=O)N[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1OP([O-])(=O)OP([O-])(=O)OC[C@H]2OC([C@H](O)[C@@H]2O)N3C=CC(=O)NC3=O
InChI
1S/C17H27N3O17P2.2Na/c1-6(22)18-10-13(26)11(24)7(4-21)35-16(10)36-39(31,32)37-38(29,30)33-5-8-12(25)14(27)15(34-8)20-3-2-9(23)19-17(20)28;;/h2-3,7-8,10-16,21,24-27H,4-5H2,1H3,(H,18,22)(H,29,30)(H,31,32)(H,19,23,28);;/q;2*+1/p-2/t7-,8-,10-,11+,12-,13-,14-,15?,16?;;/m1../s1
InChI key
HXWKMJZFIJNGES-QCVFHWOISA-L
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
The presence of multiple functional groups and stereocenters in complex carbohydrates makes them challenging targets for the organic chemist.
Glycosyltransferases were initially considered to be specific for a single glycosyl donor and acceptor, which led to the one enzyme-one linkage concept. Subsequent observations have refuted the theory of absolute enzymatic specificity by describing the transfer of analogs of some nucleoside mono- or diphosphate sugar donors.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service