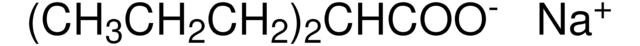A5513
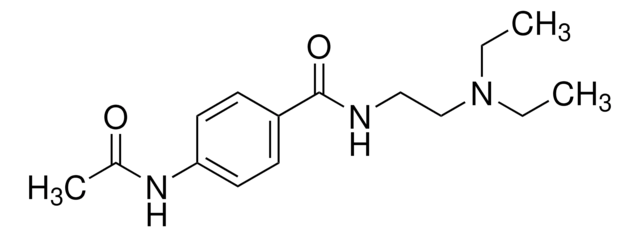
N-Acetylprocainamide hydrochloride
≥99% (HPLC), powder
Synonym(s):
N-Acetylnovocainamide hydrochloride, Acecainide hydrochloride, NAPA
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
4-(CH3CONH)C6H4CONHCH2CH2N(C2H5)2·HCl
CAS Number:
Molecular Weight:
313.82
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.77
Recommended Products
Assay
≥99% (HPLC)
form
powder
mp
184-186 °C (lit.)
solubility
H2O: 50 mg/mL
storage temp.
−20°C
SMILES string
Cl[H].CCN(CC)CCNC(=O)c1ccc(NC(C)=O)cc1
InChI
1S/C15H23N3O2.ClH/c1-4-18(5-2)11-10-16-15(20)13-6-8-14(9-7-13)17-12(3)19;/h6-9H,4-5,10-11H2,1-3H3,(H,16,20)(H,17,19);1H
InChI key
IYEWBJUCJHKLHD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
N-Acetylprocainamide hydrochloride may be used:
- as an internal standard for spiking plasma samples for ultra-high-pressure liquid chromatography coupled with a diode array detector (UHPLC-DAD) analysis
- to test its relaxant effect on tracheal smooth muscle tissue preparations
- in preparation of complexes with N-acetyl-L-tyrosine methyl ester and N-acetyl-L-phenylalanine methyl ester for studying intermolecular interactions using nuclear magnetic resonance (NMR) spectroscopy studies
N-Acetylprocainamide hydrochloride is a class III antiarrhythmic compound. N-Acetylprocainamide hydrochloride has been used in a study to determine the disposition of procainamide and N-acetylprocainamide in protein-calorie malnutrition. N-Acetylprocainamide hydrochloride has also been used to study pharmacokinetics of procainamide and N-acetylprocainamide in rats.
Biochem/physiol Actions
N-acetyltransferase II in liver catalyzes the conversion of procainamide to N-acetylprocainamide (NAPA).
Class III antiarrhythmic acting on potassium currents.
Class III antiarrhythmic. Increases the duration of the action potential by decreasing the delayed outward potassium current, slightly decreasing the calcium current, and slightly depressing the inward rectifier potassium current. This is the active metabolite of procainamide that does not induce systemic lupus erythematosus.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
B L Kamath et al.
Journal of pharmaceutical sciences, 70(3), 299-302 (1981-03-01)
The pharmacokinetics of distribution and elimination of procainamide and its major metabolite, N-actylprocainamide, were studied in rats. Eight rats were selected randomly, and each received intravenously 14C-labeled procainamide hydrochloride (75 mg/kg) or 14C-labeled N-acetylprocainamide hydrochloride (86 mg/kg) according to a
D Jung et al.
Drug metabolism and disposition: the biological fate of chemicals, 13(3), 359-363 (1985-05-01)
The influence of dietary protein deficiency on the disposition of procainamide (PA) and its major metabolite, N-acetylprocainamide (NAPA) was investigated in male Sprague-Dawley rats fed for 4 weeks on a 23 (control) or a 5% (low) protein diet ad libitum.
The evidence for complex formation between N-acetyl-l-tyrosine methyl ester and N-acetylprocainamide hydrochloride using NMR spectroscopy
Janik A, et al.
Structural Chemistry, 20(4), 699-707 (2009)
Larry A Bauer et al.
Antimicrobial agents and chemotherapy, 49(4), 1649-1651 (2005-03-29)
Ten healthy adults participated in a randomized, crossover drug interaction study testing procainamide only, procainamide plus levofloxacin, and procainamide plus ciprofloxacin. During levofloxacin therapy, most procainamide and N-acetylprocainamide (NAPA) pharmacokinetic parameters, including decreased renal clearances and renal clearance/creatinine clearance ratios
M Boucher et al.
Journal of autonomic pharmacology, 18(2), 83-87 (1998-09-08)
1. The cardiac anticholinergic effects of procainamide (1 mg kg(-1) min(-1)) and its N-acetylated metabolite (NAPA) at equimolar dose (1.16 mg kg(-1) min(-1)) were studied using in vivo experimental pharmacological and in vitro radioligand binding studies. 2. Procainamide and NAPA
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service