C116602
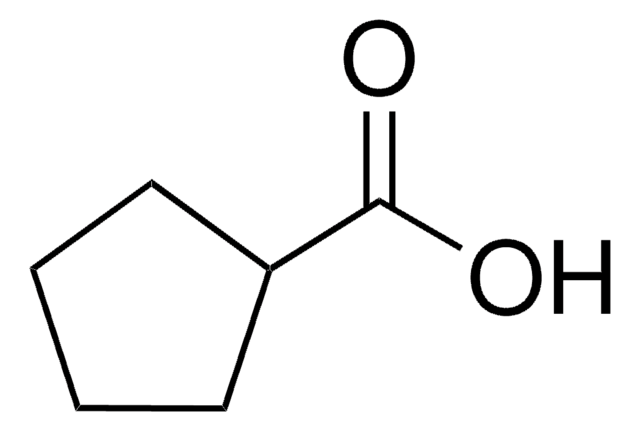
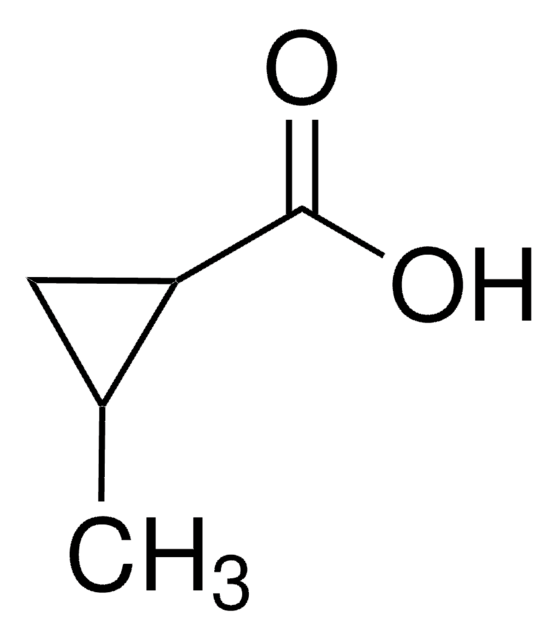
Cyclopropanecarboxylic acid
95%
Synonym(s):
CPC-acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
C3H5CO2H
CAS Number:
Molecular Weight:
86.09
Beilstein:
969839
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
95%
form
liquid
refractive index
n20/D 1.438 (lit.)
bp
182-184 °C (lit.)
mp
14-17 °C (lit.)
density
1.081 g/mL at 25 °C (lit.)
storage temp.
2-8°C
SMILES string
OC(=O)C1CC1
InChI
1S/C4H6O2/c5-4(6)3-1-2-3/h3H,1-2H2,(H,5,6)
InChI key
YMGUBTXCNDTFJI-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
161.6 °F - closed cup
Flash Point(C)
72 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mei-Xiang Wang et al.
The Journal of organic chemistry, 68(2), 621-624 (2003-01-18)
Biotransformations of differently configured 2,2-dimethyl-3-substitued-cyclopropanecarbonitriles were studied using a nitrile hydratase/amidase-containing Rhodococcus sp. AJ270 whole-cell catalyst under very mild conditions. Although all of the cis-3-aryl-2,2-dimethylcyclopropanecarbonitriles appeared inert toward the biocatalyst, a number of racemic trans-isomers efficiently underwent a highly enantioselective
M V Clos et al.
British journal of pharmacology, 118(4), 901-904 (1996-06-01)
1. The effect of 1-aminocyclopropanecarboxylic acid (ACPC), a partial agonist at the glycine site of the N-methyl-D-aspartate (NMDA) receptor complex that exhibits neuroprotective, anxiolytic and antidepressant-like actions, was investigated in a functional assay for presynaptic NMDA receptors. 2. NMDA (100
Hiroaki Yasukochi et al.
Organic & biomolecular chemistry, 6(3), 540-547 (2008-01-26)
We performed an efficient practical and systematic optical resolution method for gem-dihalo- and monohalocyclopropanecarboxylic acids and utilizing chiral 1,1'-binaphthol monomethyl ether (R)- as the key auxiliary. Direct esterification of with (R)- gave two 1R- and 1S-diastereomeric esters with marked different
H M Badawi et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 71(4), 1540-1546 (2008-07-05)
The structural stability and internal rotations in cyclopropanecarboxylic acid and cyclopropanecarboxamide were investigated by the DFT-B3LYP and the ab initio MP2 calculations using 6-311G** and 6-311+G** basis sets. The computations were extended to the MP4//MP2/6-311G** and CCSD(T)//MP2/6-311G** single-point calculations. From
Tetsuo Toraya et al.
Applied and environmental microbiology, 70(1), 224-228 (2004-01-09)
A new strain isolated from soil utilizes cyclopropanecarboxylate as the sole source of carbon and energy and was identified as Rhodococcus rhodochrous (H. Nishihara, Y. Ochi, H. Nakano, M. Ando, and T. Toraya, J. Ferment. Bioeng. 80:400-402, 1995). A novel
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service