674788
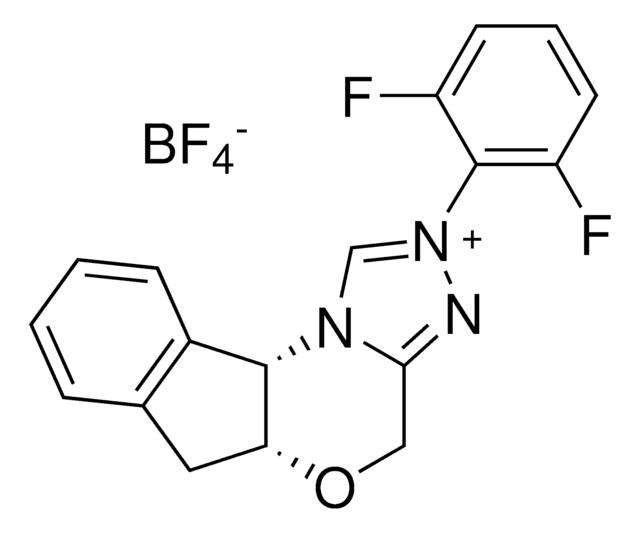
5a(R),10b(S)-5a,10b-Dihydro-2-(pentafluorophenyl)-4H,6H-indeno[2,1-b][1,2,4]triazolo[4,3-d][1,4]oxazinium tetrafluoroborate
97%
Synonym(s):
2-Pentafluorophenyl-6,10b-dihydro-4H,5aH-5-oxo-3,10c-diaza-2-azoniacyclopenta[c]fluorine tetrafluoroborate
About This Item
Recommended Products
Assay
97%
form
solid
mp
233-237 °C
storage temp.
2-8°C
SMILES string
F[B-](F)(F)F.Fc1c(F)c(F)c(c(F)c1F)-[n+]2c[n@@H]3[C@@H]4[C@@H](Cc5ccccc45)OCc3n2
InChI
1S/C18H11F5N3O.BF4/c19-12-13(20)15(22)18(16(23)14(12)21)26-7-25-11(24-26)6-27-10-5-8-3-1-2-4-9(8)17(10)25;2-1(3,4)5/h1-4,7,10,17H,5-6H2;/q+1;-1/t10-,17+;/m1./s1
InChI key
CPCMDOOTVHDRTM-CVJFODCESA-N
Application
- Benzoin reactions of aldehydes and total synthesis of a natural product named isodarparvinol B.
- Synthesis of spirocyclic oxindole-dihydropyranones by reacting α-bromo-α,β-unsaturated aldehydes with isatin derivatives.
- Synthesis of 1,4-dicarbonyl compounds through intramolecular Stetter reaction.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1 - Skin Corr. 1B
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Rovis has demonstrated that triazolium salt in the presence of a base can act as an N-heterocyclic carbene organocatalyst in highly enantioselective intramolecular Stetter reactions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![6,7-Dihydro-2-pentafluorophenyl-5H-pyrrolo[2,1-c]-1,2,4-triazolium tetrafluoroborate 97%](/deepweb/assets/sigmaaldrich/product/structures/338/297/58bedd1e-fa2f-4f2d-92eb-c61b56af8ded/640/58bedd1e-fa2f-4f2d-92eb-c61b56af8ded.png)
![(5aR,10bS)-5a,10b-Dihydro-2-(2,4,6-trimethylphenyl)-4H,6H-indeno[2,1-b]-1,2,4-triazolo[4,3-d]-1,4-oxazinium chloride monohydrate 93%](/deepweb/assets/sigmaaldrich/product/structures/104/483/183b49bc-426f-411b-8d11-71bbd4b81022/640/183b49bc-426f-411b-8d11-71bbd4b81022.png)

![2-Mesityl-2,5,6,7-tetrahydropyrrolo[2,1-c][1,2,4]triazol-4-ium chloride 97%](/deepweb/assets/sigmaaldrich/product/structures/267/516/0a2e9bce-0442-44c8-b912-3f3eeae583cf/640/0a2e9bce-0442-44c8-b912-3f3eeae583cf.png)
![(5aS,10bR)-5a,10b-Dihydro-2-mesityl-4H,6H-indeno[2,1-b]-1,2,4-triazolo[4,3-d]-1,4-oxazinium chloride](/deepweb/assets/sigmaaldrich/product/structures/226/284/a4e3161c-6ede-440f-8902-6af8e576a3ab/640/a4e3161c-6ede-440f-8902-6af8e576a3ab.png)
![(S)-5-Benzyl-2-mesityl-6,6-dimethyl-6,8-dihydro-5H-[1,2,4]triazolo[3,4-c][1,4]oxazin-2-ium tetrafluoroborate](/deepweb/assets/sigmaaldrich/product/structures/264/074/1f7b927e-bdc3-4352-9a96-ff81396c1618/640/1f7b927e-bdc3-4352-9a96-ff81396c1618.png)
![(5R,6S)-2-Mesityl-5,6-diphenyl-6,8-dihydro-5H-[1,2,4]triazolo[3,4-c][1,4]oxazin-2-ium tetrafluoroborate 97%](/deepweb/assets/sigmaaldrich/product/structures/219/182/9bfa803e-8970-4dd1-9cbf-9ebca2f74da2/640/9bfa803e-8970-4dd1-9cbf-9ebca2f74da2.png)