419443
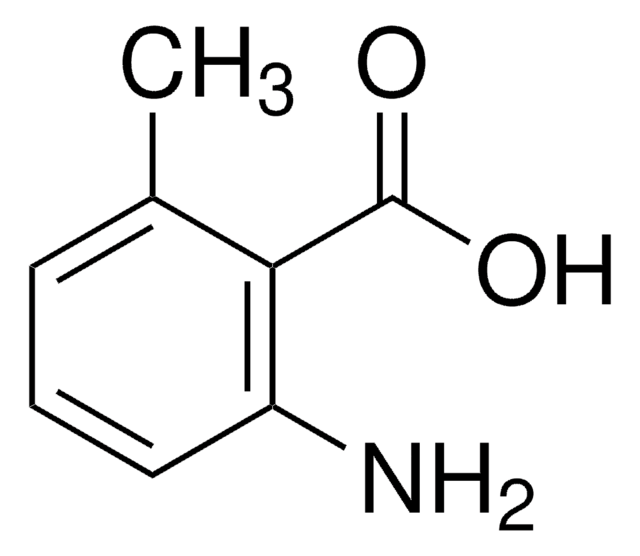
2-Amino-5-methylbenzoic acid
99%
Synonym(s):
5-Methylanthranilic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
H2NC6H3(CH3)CO2H
CAS Number:
Molecular Weight:
151.16
Beilstein:
1101527
EC Number:
MDL number:
UNSPSC Code:
12352106
eCl@ss:
32160406
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
reaction suitability
reaction type: solution phase peptide synthesis
mp
175 °C (dec.) (lit.)
application(s)
peptide synthesis
SMILES string
Cc1ccc(N)c(c1)C(O)=O
InChI
1S/C8H9NO2/c1-5-2-3-7(9)6(4-5)8(10)11/h2-4H,9H2,1H3,(H,10,11)
InChI key
NBUUUJWWOARGNW-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
2-Amino-5-methylbenzoic acid also known as 5-methylanthranilic acid, is often used in the solution-phase peptide synthesis.
Application
2-Amino-5-methylbenzoic acid can be used to synthesize quinazolinedione.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
João Daniel Santos Fernandes et al.
PloS one, 10(7), e0132369-e0132369 (2015-07-15)
Metabolic diversity is an important factor during microbial adaptation to different environments. Among metabolic processes, amino acid biosynthesis has been demonstrated to be relevant for survival for many microbial pathogens, whereas the association between pathogenesis and amino acid uptake and
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service