추천 제품
Quality Level
분석
≥98% (HPLC)
형태
solid
광학 활성
[α]28/D −2.2°, c = 0.9 in methanol(lit.)
색상
white
solubility
DMSO: >20 mg/mL
SMILES string
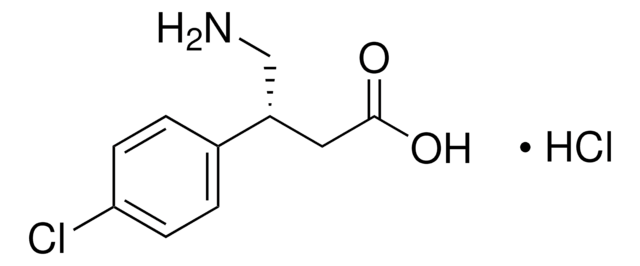
Cl[H].NC[C@@H](CC(O)=O)c1ccc(Cl)cc1
InChI
1S/C10H12ClNO2.ClH/c11-9-3-1-7(2-4-9)8(6-12)5-10(13)14;/h1-4,8H,5-6,12H2,(H,13,14);1H/t8-;/m1./s1
InChI key
WMNUVYYLMCMHLU-DDWIOCJRSA-N
생화학적/생리학적 작용
S(−)-Baclofen hydrochloride is a less active enantiomer of baclofen.
기타 정보
Same enantiomer as S(+)-baclofen free base.
Storage Class Code
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
시험 성적서(COA)
제품의 로트/배치 번호를 입력하여 시험 성적서(COA)을 검색하십시오. 로트 및 배치 번호는 제품 라벨에 있는 ‘로트’ 또는 ‘배치’라는 용어 뒤에서 찾을 수 있습니다.
R Paredes et al.
Psychopharmacology, 97(3), 358-364 (1989-01-01)
The behavior effects of racemic baclofen and the R and S enantiomers were studied in order to determine whether the stereospecificity found in receptor binding studies also applies to the behavioral actions of the drug. Racemic and R-baclofen inhibited sexual
Y G Hong et al.
European journal of pharmacology, 196(3), 267-275 (1991-04-24)
In a previous study it was found that i.t. administration of L-baclofen decreased arterial pressure and heart rate while D-baclofen differentially increased arterial pressure. The objective of the present study was to determine which of these effects was blocked by
E Falch et al.
Journal of neurochemistry, 47(3), 898-903 (1986-09-01)
The affinities of a number of analogues of gamma-aminobutyric acid (GABA) for GABAA and GABAB receptor sites and GABA uptake were studied using rat brain membrane preparations. Studies on the (S)-(+)- and (R)-(-)-isomers of baclofen, 3-hydroxy-4-aminobutyric acid (3-OH-GABA), and 4,5-dihydromuscimol
B Witczuk et al.
Polish journal of pharmacology and pharmacy, 32(2), 187-196 (1980-03-01)
Racemic 3-(p-chlorophenyl)-4-aminobutanoic acid was resolved into enantiomers and their absolute configuration determined. Pharmacological activity of hydrochlorides of the racemic acid and its enantiomers has been determined. The R(+) enantiomer was found to be 4.2-9.2-fold as effective as the S(-) one
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.