추천 제품
Grade
pharmaceutical primary standard
API family
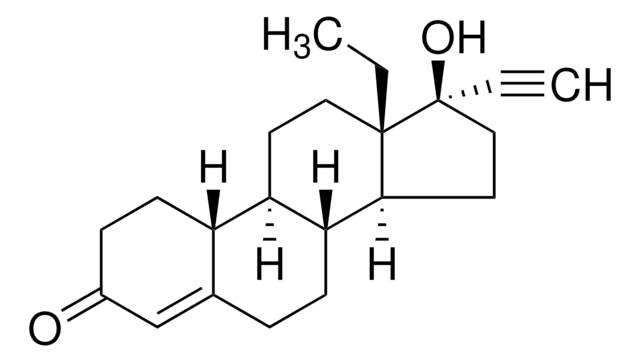
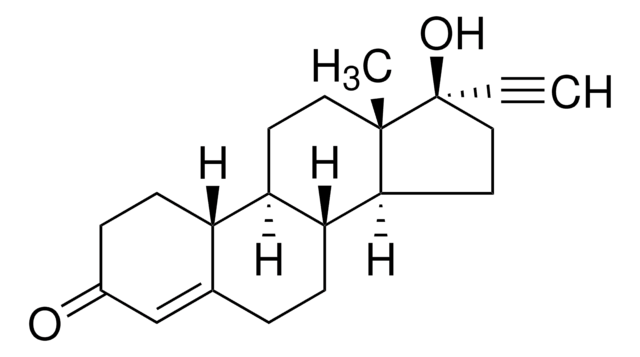
levonorgestrel
제조업체/상표
EDQM
응용 분야
pharmaceutical (small molecule)
형식
neat
InChI
1S/C21H28O2/c1-3-20-11-9-17-16-8-6-15(22)13-14(16)5-7-18(17)19(20)10-12-21(20,23)4-2/h2,17-19,23H,3,5-13H2,1H3/t17-,18-,19+,20+,21+/m1/s1
InChI key
QCMLVDXPKCBMFQ-MJCUULBUSA-N
일반 설명
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the Issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
애플리케이션
Levonorgestrel impurity B EP Reference standard, intended for use in laboratory tests only as specifically prescribed in the European Pharmacopoeia.
포장
The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity, please visit the EDQM reference substance catalogue.
기타 정보
Sales restrictions may apply.
관련 제품
제품 번호
설명
가격
가장 최신 버전 중 하나를 선택하세요:
Cheng Li et al.
The Journal of pathology, 248(1), 77-87 (2019-01-12)
Previous studies revealed the increasing risk of tubal pregnancy following failure of levonorgestrel (LNG)-induced emergency contraception, which was attributed to the reduced ciliary motility in response to LNG. However, understanding of the mechanism of LNG-induced reduction in the ciliary beat
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.