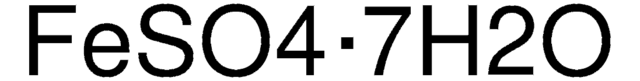930989
Magnesium chloride hexahydrate
99.999% (trace metals basis)
동의어(들):
Magnesium dichloride hexahydrate
로그인조직 및 계약 가격 보기
모든 사진(1)
About This Item
Linear Formula:
MgCl2 · 6H2O
CAS Number:
Molecular Weight:
203.30
MDL number:
UNSPSC 코드:
12352302
NACRES:
NA.21
solubility:
H2O: soluble (highly soluble)
acetone: slightly soluble
alcohols: slightly soluble
acetone: slightly soluble
alcohols: slightly soluble
추천 제품
Grade
for analytical purposes
Quality Level
분석
99.999% (trace metals basis)
양식
crystalline
mp
117 °C
solubility
H2O: soluble (highly soluble)
acetone: slightly soluble
alcohols: slightly soluble
SMILES string
O.O.O.O.O.O.Cl[Mg]Cl
InChI
1S/2ClH.Mg.6H2O/h2*1H;;6*1H2/q;;+2;;;;;;/p-2
InChI key
DHRRIBDTHFBPNG-UHFFFAOYSA-L
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Magnesium chloride hexahydrate is a colorless to white, crystalline salt. The salt is soluble in water and slightly soluble in ethanol, acetone, and pyridine. The hydrate loses water when heated to higher temperatures, but thermal dehydration to the anhydrous salt is not straightforward. Chemically, magnesium chloride is a weak Lewis acid. One of the most common and simplest routes to producing magnesium chloride is extraction from naturally occurring brine, for example by solar evaporation of sea water, which can be purified by solution chemistries.
애플리케이션
One major application of high-purity magnesium chloride is in the synthesis of layered double hydroxides (LDH), which are high-surface area ionic clays. LDHs are studied for water filtration, among other applications, and usually consist of magnesium and aluminum ions because of the low toxicity of these metal cations. LDHs are synthesized from chloride salts using co-precipitation reactions, ion-exchange processes, and urea methods. Another application of high-purity magnesium chloride is as a reagent to dope or substitute magnesium ions into solid-state materials and nanoparticles. For example, Mg-doping in ZnO has shown to modulate its band structure and Mg-doping in lithium nickel cobalt manganese oxide helps suppress cation mixing and enables longer cycle lifetimes for its use as a cathode material.
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 1
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.