추천 제품
양식
powder or solid
분자량
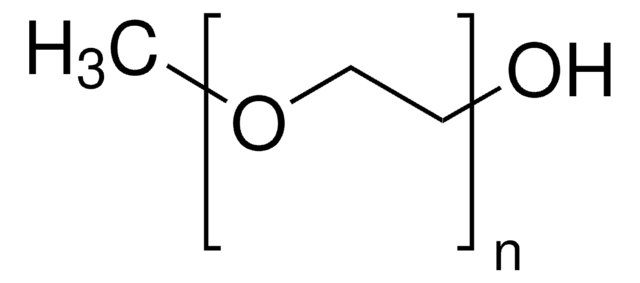
PEG average Mn 10,000
PEG ~10,000 Da
색상
off-white to pale yellow
저장 온도
2-8°C
일반 설명
Poly(ethylene glycol) bis(2-pyridyl KAT) is a homobifunctional PEG featuring terminal potassium acyltrifluoroborate reactive groups for facile, rapid functionalization. Potassium acyltrifluoroborates (KATs) are stable functional groups that undergo rapid amide-forming ligations with hydroxylamines in aqueous media, in the presence of unprotected functional groups. In addition to its compatibility, these reactions proceed relatively quickly, lending to their use with sensitive biological reagents. This conjugation reaction offers a new approach to the synthesis of complex molecules without the complication of side reactions, such protein-polymer conjugates. KATs also undergo amide or imide-forming ligations in acidic conditions when reacted with primary amines or amides, respectively, as an alternative to classical acylation chemistry. Poly(ethylene glycol) bis(2-pyridyl KAT)s have been recently used in the rapid PEGylation and dimerization of expressed, folded protiens in near equimolar conditions, demonstrating the potential for these materials in a wide variety of drug delivery applications.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
가장 최신 버전 중 하나를 선택하세요:
PEGylation and Dimerization of Expressed Proteins under Near Equimolar Conditions with Potassium 2-Pyridyl Acyltrifluoroborates.
White CJ, et al.
ACS central science (2017)
Potassium Acyltrifluoroborate (KAT) Ligations are Orthogonal to Thiol-Michael and SPAAC Reactions: Covalent Dual Immobilization of Proteins onto Synthetic PEG Hydrogels.
Mazunin D, et al.
Helvetica Chimica Acta, 100(2) (2017)
Critical evaluation and rate constants of chemoselective ligation reactions for stoichiometric conjugations in water.
Saito, et al.
ACS Chemical Biology, 10, 1026-1033 (2015)
Alberto Osuna Gálvez et al.
Journal of the American Chemical Society, 139(5), 1826-1829 (2017-01-25)
Current methods for constructing amide bonds join amines and carboxylic acids by dehydrative couplings-processes that usually require organic solvents, expensive and often dangerous coupling reagents, and masking other functional groups. Here we describe an amide formation using primary amines and
Fumito Saito et al.
ACS chemical biology, 10(4), 1026-1033 (2015-01-13)
Chemoselective ligation reactions have contributed immensely to the development of organic synthesis and chemical biology. However, the ligation of stoichiometric amounts of large molecules for applications such as protein-protein conjugates is still challenging. Conjugation reactions need to be fast enough
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.