모든 사진(1)
About This Item
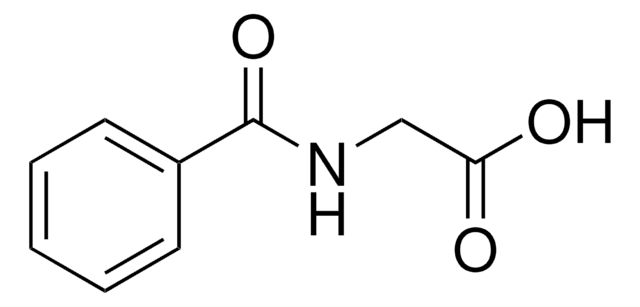
Linear Formula:
CH3C6H4CONHCH2CO2H
CAS Number:
Molecular Weight:
193.20
MDL number:
UNSPSC 코드:
12352100
PubChem Substance ID:
NACRES:
NA.22
추천 제품
Quality Level
분석
98%
mp
138-140 °C (lit.)
작용기
amide
carboxylic acid
SMILES string
Cc1cccc(c1)C(=O)NCC(O)=O
InChI
1S/C10H11NO3/c1-7-3-2-4-8(5-7)10(14)11-6-9(12)13/h2-5H,6H2,1H3,(H,11,14)(H,12,13)
InChI key
YKAKNMHEIJUKEX-UHFFFAOYSA-N
관련 카테고리
일반 설명
3-Methylhippuric acid is also referred as m-methyl-hippuric acid. It is major product of xylene biotransformation in urine.
애플리케이션
3-Methylhippuric acid was employed as biological marker in studies on occupational exposure to xylene (solvent).
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
개인 보호 장비
Eyeshields, Gloves, type N95 (US)
이미 열람한 고객
D de Carvalho et al.
International archives of occupational and environmental health, 63(1), 33-37 (1991-01-01)
The industrial solvents, toluene and xylene, have physicochemical properties that can be hazardous to the workers exposed. Since hippuric acid and m-methyl-hippuric acid represent the products of toluene and xylene biotransformation in urine, they are used as biological markers in
L Campbell et al.
British journal of industrial medicine, 45(2), 127-132 (1988-02-01)
In a series of experiments to investigate interactions between industrial solvents and common medications the interaction between m-xylene and aspirin was studied. As both these substances are metabolised and excreted as glycine conjugates there would possibly be competition for this
Determination of methylhippuric acid in human urine by high-performance liquid chromatography and by isotachophoresis.
J Sollenberg et al.
Journal of chromatography, 343(2), 419-423 (1985-10-11)
A Astier
Journal of chromatography, 573(2), 318-322 (1992-01-17)
A high-performance liquid chromatographic method is described for the simultaneous determination of six urinary metabolites of several aromatic chemicals: phenol (from benzene), hippuric acid (from toluene), 3-methylhippuric acid (from xylene), mandelic and phenylglyoxylic acid (from styrene) and 4-nitrophenol (from nitrobenzene).
Possible preferential metabolism of xylene isomers following occupational exposure to mixed xylenes.
M J Miller et al.
International archives of occupational and environmental health, 72(2), 89-97 (1999-04-10)
Solvent exposures commonly involve mixtures of substances or mixtures of isomers of a single solvent. These may be metabolised through common pathways, resulting in the potential for metabolic interactions. These may then lead to accumulation of solvent or metabolic intermediates
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 328014-5G | |
| 328014-1G | 4061826717141 |
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.