M2389
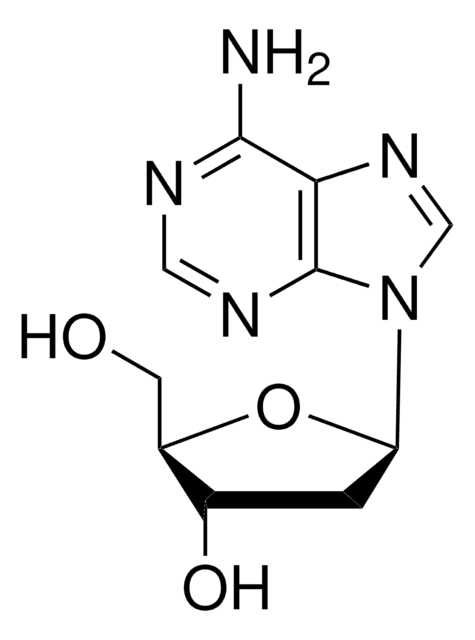
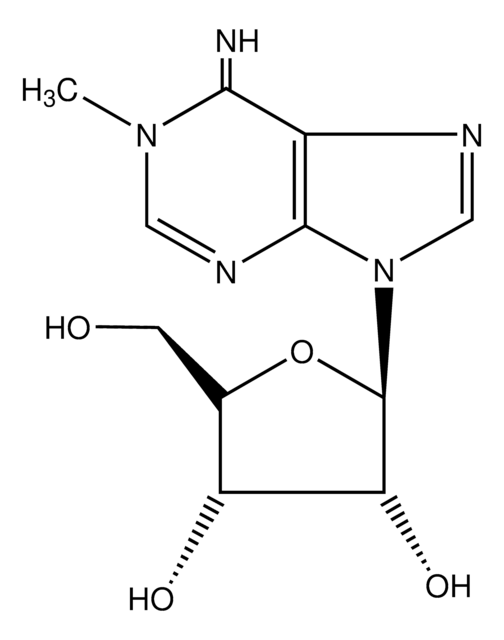
N6-Methyl-2′-deoxyadenosine
Synonym(s):
6mdA, N6-Me-dAdo, m6dA
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
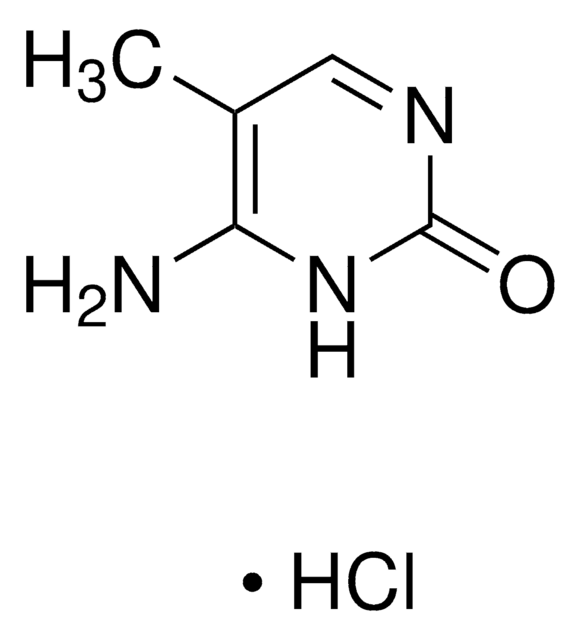
Empirical Formula (Hill Notation):
C11H15N5O3
CAS Number:
Molecular Weight:
265.27
MDL number:
UNSPSC Code:
41106305
PubChem Substance ID:
NACRES:
NA.51
Recommended Products
Assay
≥98% (TLC)
Quality Level
form
powder
solubility
acetic acid: 49.00-51.00 mg/mL, clear, colorless
storage temp.
−20°C
SMILES string
CNc1ncnc2n(cnc12)[C@H]3C[C@H](O)[C@@H](CO)O3
InChI
1S/C11H15N5O3/c1-12-10-9-11(14-4-13-10)16(5-15-9)8-2-6(18)7(3-17)19-8/h4-8,17-18H,2-3H2,1H3,(H,12,13,14)/t6-,7+,8+/m0/s1
InChI key
DYSDOYRQWBDGQQ-XLPZGREQSA-N
Application
N6-Methyl-2′-deoxyadenosine has been used as a standard to assess the levels of modified bases in genomic DNA by the ultra-high-performance liquid chromatography coupled with triple quadrupole mass spectrometry (UHPLC-QQQ-MS/MS) method. It has also been used to study its potential in promoting the proliferation oferythroid progenitor cells.
Biochem/physiol Actions
N6-Methyl-2′-deoxyadenosine (N6-Me-dAdo) is a precursor of N6-methyl 2′-deoxyadenosine 3′,5′-bisphosphate (N6MABP), a competitive and selective inhibitor of P2Y1 receptors. It is located at the transcription start site and plays a role in increased gene expression in Chlamydomonas reindardtii. N6-Me-dAdo augments the within transposable elements and is present at higher levels during the development of Drosophila. It is involved in the regulation of gene transcription, DNA repair, and replication in prokaryotes and protists. N6-Me-dAdo complemented with gene bodies might induce gene expression in human cells.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
J C Rogers et al.
The Plant journal : for cell and molecular biology, 7(2), 221-233 (1995-02-01)
Methylation of deoxycytosine residues in plant nuclear DNA at CpG dinucleotides is generally assumed to suppress transcription, while deoxyadenosine methylation on recombinant reporter gene constructs transiently introduced into plant cells increases expression by uncharacterized mechanisms. A particle bombardment transient expression
Kelly M Zatopek et al.
DNA repair, 80, 36-44 (2019-06-28)
RAre DAmage and Repair sequencing (RADAR-seq) is a highly adaptable sequencing method that enables the identification and detection of rare DNA damage events for a wide variety of DNA lesions at single-molecule resolution on a genome-wide scale. In RADAR-seq, DNA
Mariangela Chisari et al.
Molecular neurobiology, 54(6), 4081-4093 (2016-06-20)
Astrocytes regulate neuronal activity and blood brain barrier through tiny plasma membrane branches or astrocytic processes (APs) making contact with synapses and brain vessels. Several transmitters released by astrocytes and exerting their action on several receptor classes expressed by astrocytes
J J Solomon et al.
Chemico-biological interactions, 51(2), 167-190 (1984-09-15)
Reaction of the rodent carcinogen acrylonitrile (AN) at pH 5.0 and/or pH 7.0 for 10 and/or 40 days with 2'-deoxyadenosine (dAdo), 2'-deoxycytidine (dCyd), 2'-deoxyguanosine (dGuo), 2'-deoxyinosine (dIno), N6-methyl-2'-deoxyadenosine (N6-Me-dAdo) and thymidine (dThd) resulted in the formation of cyanoethyl and carboxyethyl
J L Boyer et al.
British journal of pharmacology, 124(1), 1-3 (1998-06-18)
The antagonist activity of N6-methyl 2'-deoxyadenosine 3',5'-bisphosphate (N6MABP) has been examined at the phospholipase C-coupled P2Y1 receptor of turkey erythrocyte membranes. N6MABP antagonized 2MeSATP-stimulated inositol phosphate hydrolysis with a potency approximately 20 fold greater than the previously studied parent molecule
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service