V2002
Vancomycin hydrochloride from Streptomyces orientalis
≥900 μg per mg (as vancomycin base)
Synonym(s):
Vancomycin, Vancomycin HCL
About This Item
Recommended Products
biological source
Streptomyces orientalis
Quality Level
form
powder
storage condition
(Keep container tightly closed in a dry and well-ventilated place.)
concentration
≥900 μg/mg (as vancomycin base)
color
, off-white to brown or White to orange-brown
antibiotic activity spectrum
Gram-positive bacteria
Mode of action
cell wall synthesis | interferes
storage temp.
2-8°C
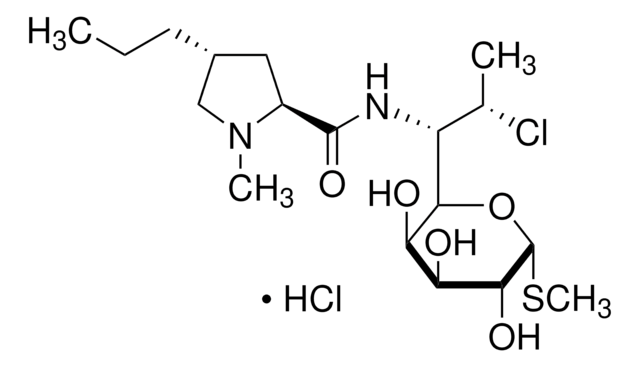
SMILES string
Cl[H].CO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@@H]1O[C@H]2C[C@](C)(N)[C@@H](O)[C@@H](C)O2.CN[C@H](CC(C)C)C(=O)NC3[C@H](O)c4ccc(Oc5cc6Oc7ccc(cc7Cl)[C@@H](O)[C@H]8NC(=O)[C@H](NC(=O)[C@H](NC(=O)[C@H](CC(N)=O)NC3=O)c(c5)c6)c9ccc(O)c(c9)-c%10c(O)cc(O)cc%10[C@@H](NC8=O)C(O)=O)c(Cl)c4
InChI
1S/C66H75Cl2N9O24.ClH/c1-23(2)12-34(71-5)58(88)76-49-51(83)26-7-10-38(32(67)14-26)97-40-16-28-17-41(55(40)101-65-56(54(86)53(85)42(22-78)99-65)100-44-21-66(4,70)57(87)24(3)96-44)98-39-11-8-27(15-33(39)68)52(84)50-63(93)75-48(64(94)95)31-18-29(79)19-37(81)45(31)30-13-25(6-9-36(30)80)46(60(90)77-50)74-61(91)47(28)73-59(89)35(20-43(69)82)72-62(49)92;/h6-11,13-19,23-24,34-35,42,44,46-54,56-57,65,71,78-81,83-87H,12,20-22,70H2,1-5H3,(H2,69,82)(H,72,92)(H,73,89)(H,74,91)(H,75,93)(H,76,88)(H,77,90)(H,94,95);1H/t24-,34+,35-,42+,44-,46+,47+,48-,49+,50-,51+,52+,53+,54-,56+,57+,65-,66-;/m0./s1
InChI key
LCTORFDMHNKUSG-XTTLPDOESA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- in the research the effects of antibiotic-induced depletion of Firmicutes and Bacteroidetes on dysregulation of energy homeostasis in obesity
- in the research of non-O157 Shiga toxin-producing Escherichia coli Isolates from Bovine Farms
- to research the antimicrobial susceptibility of Bifidobacterium strains in various organisms
Biochem/physiol Actions
Antimicrobial Spectrum: Active against Gram-positive bacteria
Features and Benefits
- Effective against a wide range of Gram-positive bacteria, including MRSA
- Commonly used in Cell Biology and Biochemical applications
Packaging
Other Notes
comparable product
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
V2002-BULK:
V2002-250MG:
V2002-VAR:
V2002-100MG:
V2002-1G:
V2002-5G:
V2002-5G-PW:
V2002-100G:
V2002-100G-PW:
V2002-250MG-PW:
V2002-1G-PW:
V2002-100MG-PW:
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Inhibition of Cell Wall Biosynthesis by Antibiotics
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service