SAE0097
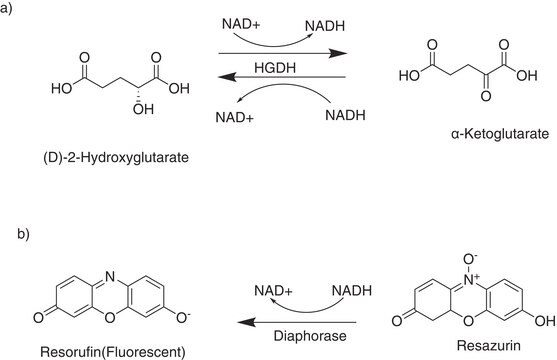
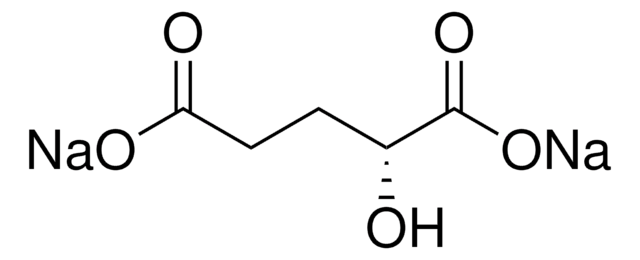
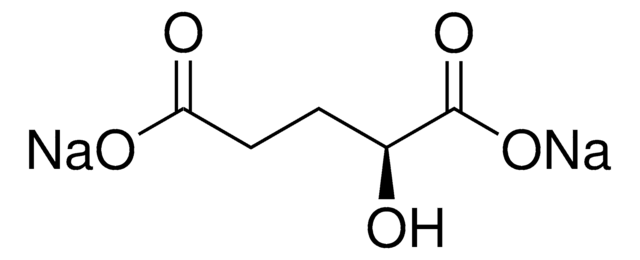
D-2-Hydroxyglutarate Dehydrogenase (D2HGDH) from Acidaminococcus fermentans
recombinant, expressed in E. coli, aqueous solution
Synonym(s):
D2HGDH, HGDH, L-2-hydroxyglutarate dehydrogenase
About This Item
Recommended Products
recombinant
expressed in E. coli
Assay
≥95% (SDS-PAGE)
form
aqueous solution
specific activity
≥1000 units/mg protein
UniProt accession no.
shipped in
wet ice
storage temp.
−20°C
General description
The crystal structure of D2HGDH from Acidaminococcus fermentans has been reported. D2HGDH from Acidaminococcus fermentans has been used in several enzymatic assays, such as:
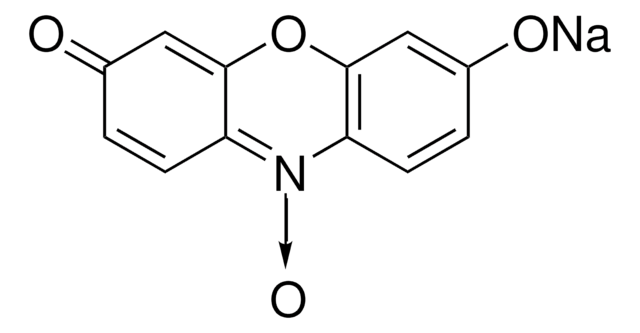
- A continuous spectrophotometric assay to measure the activity of aminotransferases, based on the transamination of a keto compound and L-glutamate, which yields a corresponding amino compound and 2-oxoglutarate.
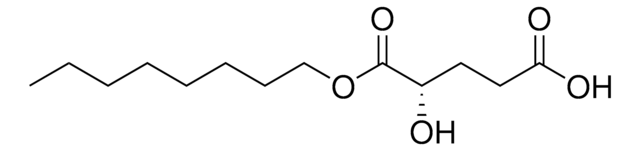
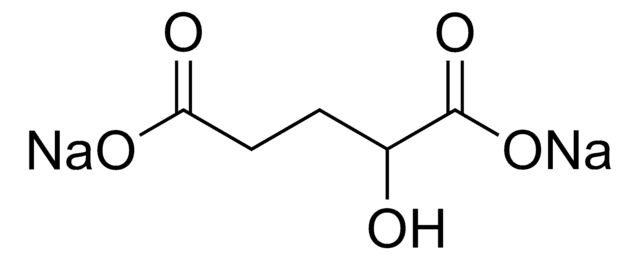
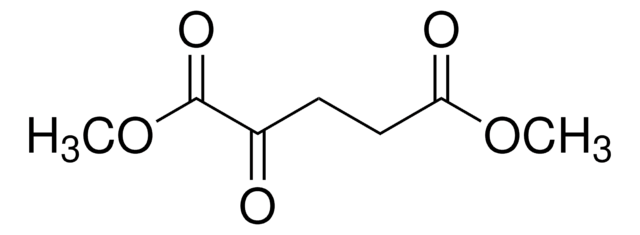
- Determination of D2HG levels in biological fluids such as serum, urine, cell culture supernatants, and cell or tissue lysates.
- A coupled assay system to measure branched-chain amino acid aminotransferase activity.
Unit Definition
Preparation Note
Legal Information
Storage Class Code
10 - Combustible liquids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
SAE0097-BULK:
SAE0097-50UG-PW:
SAE0097-VAR:
SAE0097-50UG:
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service