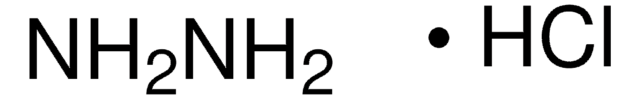225819
Hydrazine hydrate
reagent grade, N2H4 50-60 %
Synonym(s):
Hydrazinium hydroxide
About This Item
Recommended Products
grade
reagent grade
Quality Level
vapor density
>1 (vs air)
vapor pressure
5 mmHg ( 25 °C)
form
liquid
composition
Degree of hydration, ~1.5
N2H4, 50-60%
expl. lim.
99.99 %
concentration
50.0-60.0% (%H4N2 by Na2S2O3 basis, titration)
density
1.029 g/mL at 25 °C (lit.)
SMILES string
NN.[H]O[H]
InChI
1S/H4N2.H2O/c1-2;/h1-2H2;1H2
InChI key
IKDUDTNKRLTJSI-UHFFFAOYSA-N
General description
Application
- Preparation of silver nanoparticles.
- Transformation of monosubstituted nitrobenzene derivatives to the corresponding anilines.
- Along with graphite for the conversion of nitro compounds (aromatic and aliphatic) to the amino compounds.
- 3-(2-Benzyloxy-6-hydroxyphenyl)-5-styrylpyrazoles by reacting with 5-benzyloxy-2-styrylchromones.
- 3,5-Diphenyl-2-pyrazoline derivatives by reacting with 1,3-diphenyl-2-propen-1-one.
- 3′-Aryl-1,2,3,4,4′,5′-hexahydrospiro[quinoxalin-2,5′-pyrazol]-3-ones by reacting with 3-arylacylidene-3,4-dihydroquinoxalin-2(1H)-ones.
- It may also be used in the catalytic reduction of nitroarenes to aromatic amines.
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 2 Inhalation - Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1B - Eye Dam. 1 - Flam. Liq. 3 - Skin Corr. 1B - Skin Sens. 1
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
204.8 °F - closed cup
Flash Point(C)
96 °C - closed cup
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
PDSCL
Deleterious substance
PRTR
Class I Designated Chemical Substances
FSL
Group 4: Flammable liquids
Type 3 petroleums
Hazardous rank III
Water insoluble liquid
ISHL Indicated Name
Substances Subject to be Indicated Names
ISHL Notified Names
Substances Subject to be Notified Names
JAN Code
225819-1KG:4548173119328
225819-500ML:
225819-VAR:
225819-50G:4548173119342
225819-100ML:
225819-250G:4548173119335
225819-BULK:
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Professor Aran (Claremont University, USA) thoroughly discusses the engineering of graphene based materials through careful functionalization of graphene oxide, a solution processable form of graphene.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service